The activity of Ixophilin might, in part, be instrumental in keeping the blood stored in the tick gut in a fluid state, and thus allow nutrient uptake by pinocytosis-an important aspect of blood meal digestion in ticks. Immunity against Ixophilin significantly delayed feeding time, and decreased engorgement weights, but did not abolish feeding. Interestingly, rIxophilin-immunized mice challenged with B. burgdorferi-infected nymphs, showed increased Borrelia burden in the murine host, although there was no impact on Borrelia burden in the nymphal guts and salivary glands. Presumably, the longer feeding period might have increased the window of time for pathogen transmission to occur. The tick gut may encode more than one anticoagulant that may compensate for loss of Ixophilin function, and allow feeding to proceed, and emphasizes the need to simultaneously target all predominant members of the anticoagulome to efficiently block tick feeding. Our observations suggest stage-specific and tissue-specific expression of Ixophilin, and underscore a critical role for the tick gut in keeping the blood meal fluid. Unlike the salivary anticoagulants, the gut-specific anticoagulants are not directly exposed to host immune pressure, and may not have evolved to circumvent host immune responses. The gut “anticoagulome” might therefore be a tractable subset, and could be targeted to disrupt tick feeding, and consequent pathogen transmission. The choice of MK-4827 detection technology is another critical aspect that must be determined with awareness of the limitations associated with each method, e.g. the inner filter effect for fluorescence technologies, high substrate conversion requirement for fluorescence polarization, interference with test compounds, detection limits, and the linear range of signal responses. In HTS for enzyme inhibitors a central concern is to design an assay with a high signal-to-background ratio, and to determine an observation window that provides appropriate separation in read-out between hits and the control samples. To achieve an assay with a good Z-factor, 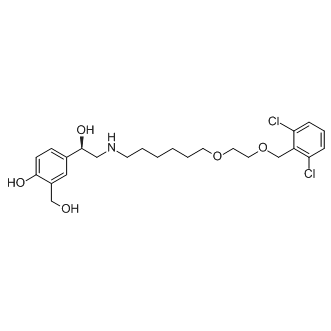 experimental noise should be minimized while maximizing the S/B ratio. A common experimental condition in HTS for enzyme inhibitors is to use low substrate concentrations to avoid saturation of the active site, which would risk missing competitive inhibitors. With low substrate concentrations it often becomes necessary to allow reactions to proceed until a large proportion of substrate becomes depleted in order to obtain sufficiently high signals. While such extended incubation times may obscure the effect of weak inhibitors, shorter incubation times give weaker signals that may adversely affect assay performance. Different modes of inhibition further complicates data WZ4002 interpretation and assay design. A further difficulty is that the underlying theory, which is based on rate-law equations for initial reaction velocity, becomes violated at extended reaction times and thus complicates data interpretation, particularly the relation between observed and true inhibitor potency. In experimental deduction of kinetic parameters, the initial reaction rate at different substrate concentrations is measured and data obtained fitted to the Michaelis-Menten rate law equation. In practice, initial reaction rates can only be approximated since the real measurable quantity represents a concentration at a given time-point.
experimental noise should be minimized while maximizing the S/B ratio. A common experimental condition in HTS for enzyme inhibitors is to use low substrate concentrations to avoid saturation of the active site, which would risk missing competitive inhibitors. With low substrate concentrations it often becomes necessary to allow reactions to proceed until a large proportion of substrate becomes depleted in order to obtain sufficiently high signals. While such extended incubation times may obscure the effect of weak inhibitors, shorter incubation times give weaker signals that may adversely affect assay performance. Different modes of inhibition further complicates data WZ4002 interpretation and assay design. A further difficulty is that the underlying theory, which is based on rate-law equations for initial reaction velocity, becomes violated at extended reaction times and thus complicates data interpretation, particularly the relation between observed and true inhibitor potency. In experimental deduction of kinetic parameters, the initial reaction rate at different substrate concentrations is measured and data obtained fitted to the Michaelis-Menten rate law equation. In practice, initial reaction rates can only be approximated since the real measurable quantity represents a concentration at a given time-point.
Author: screening library
Development of solid tumours in mice injected subcutaneously with a human ovarian cancer cell line
Increased the median survival time of mice in a human ovarian ascites model. In this communication we present biochemical, biophysical and structural characterization of 9a in its complexes with XIAP-BIR3, XIAP-BIR2BIR3 and cIAP1-BIR3. In particular, we report data on compound 9a binding to different BIR domains through analytical gel filtration and small angle X-ray scattering. Moreover, we present the crystal structures of cIAP1-BIR3 and XIAP-BIR3 domains in the presence of 9a, describing the molecular details of divalent Smac-mimetic recognition. Taken together, all the experimental evidences here reported suggest that 9a is one of the most powerful divalent Smac-mimetics known to date; the structural analysis of its recognition patterns, here presented, is the basis for further optimization in terms of target affinity and bioavailability. Compound 9a is a tail-tail homodimeric divalent Smac-mimetic that was rationally designed, together with nineteen other divalent compounds, to study how bifunctional inhibitors can bind and distinguish between XIAPand cIAPs-BIR2BIR3 domains. Among these, 9a showed prominent binding activity to BIR3 domains of XIAP and cIAPs, and to XIAP-BIR2BIR3, low cytotoxicity in two different cell lines, and the capability to induce activation of caspases and apoptosis. Moreover, the divalent compound proved effective in in vivo TH-302 treatments, after intraperitoneal daily administration, in two human IGROV1 ovarian cancer models, showing reduction of subcutaneous tumor growth in nude mice, and increase of the median survival time of mice in ascites model. Binding of the divalent compound to XIAP-BIR3 results in a crystal packing that differs from that observed in the crystal structures of the XIAP-BIR3 complexes with monovalent Smacmimetic compounds known to date. Notably, the crystal lattice packing is also different from that observed for XIAP-BIR3 in complex with the divalent compound-3, whose crystal asymmetric unit hosts eight BIR3 molecules and eight compound-3 molecules, each of which has one inhibitory head bound to BIR3 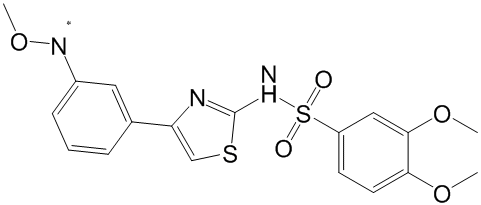 and the other devoid of any contact to the protein. Comparative SAXS analysis of XIAP-BIR2BIR3 shows that the construct in the presence of the inhibitor adopts a more compact global conformation, likely induced by 9a simultaneous binding to both BIR domains. However, ensemble analysis of free XIAP-BIR2BIR3 shows that a Nilotinib majority of the molecules adopt a compact conformation, suggesting that the two domains are transiently interacting even in the absence of 9a. Such result is also supported by a molecular dynamics simulation of XIAPBIR2BIR3 showing the conservation of an inter-domain interaction surface similar to that observed for XIAP-BIR3/BIR3/9a crystallographic dimer. A high resolution model of XIAP-BIR2BIR3/9a complex using the domain crystal structures that nicely fits SAXS data can be obtained by slightly relaxing the shape of the XIAP-BIR3/9a crystallographic dimer. In fact, a small separation of the two domains and the addition of the missing part of the structure lead to a much improved agreement with the SAXS data. In this simulated model, 9a maintains a right handed helical conformation, but with a pitch that is intermediate relative to both cIAP1-BIR3 and XIAP-BIR3. The SAXS experimental evidence of the presence of a transient interaction between XIAPBIR2BIR3, even when 9a is absent, indicates that the inhibitor may shift a preexisting equilibrium between open and closed.
and the other devoid of any contact to the protein. Comparative SAXS analysis of XIAP-BIR2BIR3 shows that the construct in the presence of the inhibitor adopts a more compact global conformation, likely induced by 9a simultaneous binding to both BIR domains. However, ensemble analysis of free XIAP-BIR2BIR3 shows that a Nilotinib majority of the molecules adopt a compact conformation, suggesting that the two domains are transiently interacting even in the absence of 9a. Such result is also supported by a molecular dynamics simulation of XIAPBIR2BIR3 showing the conservation of an inter-domain interaction surface similar to that observed for XIAP-BIR3/BIR3/9a crystallographic dimer. A high resolution model of XIAP-BIR2BIR3/9a complex using the domain crystal structures that nicely fits SAXS data can be obtained by slightly relaxing the shape of the XIAP-BIR3/9a crystallographic dimer. In fact, a small separation of the two domains and the addition of the missing part of the structure lead to a much improved agreement with the SAXS data. In this simulated model, 9a maintains a right handed helical conformation, but with a pitch that is intermediate relative to both cIAP1-BIR3 and XIAP-BIR3. The SAXS experimental evidence of the presence of a transient interaction between XIAPBIR2BIR3, even when 9a is absent, indicates that the inhibitor may shift a preexisting equilibrium between open and closed.
The flux of IQP-0410 through the ex vivo epidermal tissue was calculated physical properties
As such, the predicted complete release of IQP-0410 from the film is calculated to occur after 19.5 days from application and 50% release is calculated to occur after 9.75 days of use. The reduction in drug flux is to be expected and can be directly attributed to the difference in diffusion of IQP-0410 through epidermal tissue and a thin membrane. Most importantly, despite initial predictions that a drug with the physicochemical properties of IQP-0410 would remain in the stratum corneum, IQP-0410 was successfully released and permeated through the full thickness epidermal tissue to be collected in the basal media suggesting the potential viability of controlled zero-order delivery of IQP-0410 through the skin. While the in vitro release of IQP-0410 into dissolution media suggested an increased rate from films stored under accelerated storage conditions, this was not observed in the ex vivo permeability studies. Masitinib 790299-79-5 Transdermal drug delivery has two parts for overall drug delivery: release from formulation to the epidermis, and permeation through the skin to the underlying blood vessels. Therefore any increases in drug release rate from the film formulation will be mitigated by the diffusion of the drug through the tissue. The passive diffusion across the skin, the basis of transdermal drug 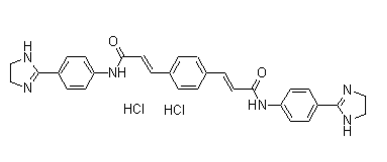 delivery, will be the limiting factor in the drug delivery and permeability. Only through external interventions to increase skin permeability, such as chemical solubility enhancers, thermal ablation, microneedles, and iontophoresis, will increased drug release from the formulations effect overall transdermal film drug delivery. To evaluate the anti-HIV efficacy of the delivered IQP-0410 through the epidermal tissue, in vitro assays in CEM-SS cells and PBMC��s against HIV-1 were performed with the collected IQP-0410 from the basal media. The concentration of IQP-0410 detected in the basal media was defined as the amount of IQP-0410 potentially bioavailable systemically and then was used to evaluate the in vitro anti-HIV efficacy and cellular toxicity performed in CEM-SS cells and PBMCs. In the basal media, IQP-0410 was collected each day, respectively, resulting in an average EC50 value in CEM-SS cells and in PBMCs over a three day application. Therefore, in conjunction with the drug recovery evaluations, we are assured that this transdermal film formulation has neither negative physicochemical nor biological effects on IQP-0410. Ultimately it is important to evaluate whether the transdermal film is capable of delivering appropriate amounts of drug into the system. Ex vivo release studies performed over three days determined that the concentration of delivered IQP-0410 after 24 hours is 5,000 fold greater than in vitro EC95 values. It is understood that the ex vivo concentrations of permeated IQP-0410 are not completely representative of potential systemic in vivo behavior; however, it can be concluded that efficacious concentrations of IQP-0410 are passing through the skin. In addition, previous product profile studies of IQP-0410 have shown that in vivo oral administration in mice results in 24% oral bioavailability with a half-life of 5.37 hours and in the presence of liver microsomes, the expected half-life in humans is 15.7 minutes. Therefore, it will be critical to avoid first pass metabolism in the liver commonly observed in oral administration to maintain therapeutic concentrations of IQP-0410 in the blood plasma by controlled delivery through the skin. One the greatest disadvantage to transdermal delivery is the possibility that a local irritation will develop at the site of administration. Irritation can be caused by the drug itself, the adhesive, or other excipients in the formulation. When applied to the skin tissue for 24 and 72 hours, the IQP-0410 transdermal films had a tissue viability of 121 �� 19.8% after a 24 hour exposure and 91.2 �� 9.71% tissue viability after 72 hours. The in vitro toxicity results from the tissue do offer a correlation in predicting clinical in vivo skin irritation. So, from the MTT TWS119 in vivo analysis of the IQP-0410 transdermal film, an in vitro ET-50 >24 hours is equivalent to the non-irritancy of 10% Tween-20. Therefore, it is expected that the IQP-0410 film formulation will result in no adverse skin irritation when administered.
delivery, will be the limiting factor in the drug delivery and permeability. Only through external interventions to increase skin permeability, such as chemical solubility enhancers, thermal ablation, microneedles, and iontophoresis, will increased drug release from the formulations effect overall transdermal film drug delivery. To evaluate the anti-HIV efficacy of the delivered IQP-0410 through the epidermal tissue, in vitro assays in CEM-SS cells and PBMC��s against HIV-1 were performed with the collected IQP-0410 from the basal media. The concentration of IQP-0410 detected in the basal media was defined as the amount of IQP-0410 potentially bioavailable systemically and then was used to evaluate the in vitro anti-HIV efficacy and cellular toxicity performed in CEM-SS cells and PBMCs. In the basal media, IQP-0410 was collected each day, respectively, resulting in an average EC50 value in CEM-SS cells and in PBMCs over a three day application. Therefore, in conjunction with the drug recovery evaluations, we are assured that this transdermal film formulation has neither negative physicochemical nor biological effects on IQP-0410. Ultimately it is important to evaluate whether the transdermal film is capable of delivering appropriate amounts of drug into the system. Ex vivo release studies performed over three days determined that the concentration of delivered IQP-0410 after 24 hours is 5,000 fold greater than in vitro EC95 values. It is understood that the ex vivo concentrations of permeated IQP-0410 are not completely representative of potential systemic in vivo behavior; however, it can be concluded that efficacious concentrations of IQP-0410 are passing through the skin. In addition, previous product profile studies of IQP-0410 have shown that in vivo oral administration in mice results in 24% oral bioavailability with a half-life of 5.37 hours and in the presence of liver microsomes, the expected half-life in humans is 15.7 minutes. Therefore, it will be critical to avoid first pass metabolism in the liver commonly observed in oral administration to maintain therapeutic concentrations of IQP-0410 in the blood plasma by controlled delivery through the skin. One the greatest disadvantage to transdermal delivery is the possibility that a local irritation will develop at the site of administration. Irritation can be caused by the drug itself, the adhesive, or other excipients in the formulation. When applied to the skin tissue for 24 and 72 hours, the IQP-0410 transdermal films had a tissue viability of 121 �� 19.8% after a 24 hour exposure and 91.2 �� 9.71% tissue viability after 72 hours. The in vitro toxicity results from the tissue do offer a correlation in predicting clinical in vivo skin irritation. So, from the MTT TWS119 in vivo analysis of the IQP-0410 transdermal film, an in vitro ET-50 >24 hours is equivalent to the non-irritancy of 10% Tween-20. Therefore, it is expected that the IQP-0410 film formulation will result in no adverse skin irritation when administered.
PXD101 with paclitaxel support consideration of use of this therapy in patients with ATC
Recently, Chan et al. also reported that PXD101 retards the growth of PTC xenograft tumors. PXD101 therefore has an ability to inhibit the growth of both well-differentiated and undifferentiated thyroid cancer in vivo. These data strengthen the possibility that PXD101 can be used to treat patients with this fatal disease. In contrast to our findings, PXD101 consistently repressed p-AKT and p-ERK in the prior study. One potential explanation of this inconsistency between two studies is the very high dose of PXD101 that was applied in SB431542 Previous study as compared with our current study. Such high doses of PXD101 are more likely to repress p-AKT and p-ERK. Our study additionally demonstrates that multiple molecular events induced by PXD101 may cause cytotoxicity, and shows the efficacy of combination therapy using PXD101 with conventional chemotherapy currently in use for anaplastic thyroid cancer. Importantly, we demonstrate synergistic effects of combination PXD101 with doxorubicin and paclitaxel, suggesting likely clinical significance in treating patients with ATC. In conclusion, PXD101 imposed significant cytotoxicity in four major histologic types of thyroid cancer. Nude mice bearing 8505C xenograft tumors demonstrated the therapeutic efficacy and safety profiles of PXD101. Importantly, PXD101 synergistically improves the therapeutic effect of doxorubicin and paclitaxel against four ATC cell lines. These favorable data support the design of future clinical trials studying the utility of PXD101 as an agent to treat patients with advanced thyroid cancer. The anthracycline antibiotic doxorubicin is used to treat a wide 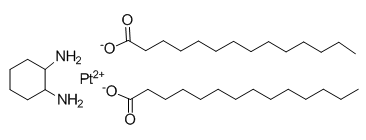 variety of cancers, but reports of its cardiotoxic properties compromises its clinical utility. The cardiotoxic effects of doxorubicin are thought to be mediated via disruption of the mitochondrial function. Previous studies have also shown doxorubicin to cause cardiotoxicity through the generation of free radicals, stimulation of lipid peroxidation and alteration and disruption of cellular membrane integrity. Arrhythmias, hypotension and depression of the contractile function are some of the acute effects of doxorubicin-induced cardiotoxicity, while chronic heart failure and dilative cardiomyopathy are more common and severe in patients who are on long term Y-27632 dihydrochloride anthracyclines treatment. Large scale clinical trials have shown that doxorubicin induced cardiotoxicity is irreversible and dose dependent. Due to advances in basic and clinical cancer research, cancer and malignancies are becoming more manageable, unfortunately the adverse cardiovascular effects of systemic anticancer agents are still a serious concern. Thus it is imperative to understand the cellular and molecular basis of doxorubicin-induced cardiotoxicity with the view to finding therapies that would offer cardioprotection without affecting its anti-tumour effects. Interventions using �� blockers, free radical scavengers, antioxidants and renin-angiotensin system inhibitors have met with limited success due, not only, to side effects but also because of their negative interactions with doxorubicin. While aiming to reduce the cardiotoxic effects of anthracyclines using adjunct therapies, it is imperative to assess the effects in cancer cell line to ascertain the clinical utility of such treatments. Interestingly, recent studies using the phosphodiesterase-5 inhibitors sildenafil or tadalafil have shown promise by showing a reduction in the cardiotoxic effects of doxorubicin without affecting its anti-cancer activity. Cell death pathways activated by doxorubicin treatment usually involve the mitochondria to initiate apoptosis or necrosis. Mitochondrial dynamics are found to play an essential role in cellular function and apoptosis. In order to maintain mitochondrial integrity and efficiency, a constant interplay between mitochondrial fission and fusion is important. Previous studies have demonstrated that upon induction of oxidative stress or ischaemia, dynamin related protein 1, a protein involved in mitochondrial fission, translocates from the cytosol to the mitochondria to execute the mitochondrial division process. This involves hydrolysing GTP, which dysregulates the balance between mitochondrial fusion and fission. Mitochondrial fission leads to cytochrome c release and activation of caspases, which can ultimately lead to cell death. Studies also reported that the dominant negative form of Drp1, DrpK38A, had the ability to inhibit mitochondrial division suggesting a regulatory role for Drp1 in mitochondria-mediated apoptosis.
variety of cancers, but reports of its cardiotoxic properties compromises its clinical utility. The cardiotoxic effects of doxorubicin are thought to be mediated via disruption of the mitochondrial function. Previous studies have also shown doxorubicin to cause cardiotoxicity through the generation of free radicals, stimulation of lipid peroxidation and alteration and disruption of cellular membrane integrity. Arrhythmias, hypotension and depression of the contractile function are some of the acute effects of doxorubicin-induced cardiotoxicity, while chronic heart failure and dilative cardiomyopathy are more common and severe in patients who are on long term Y-27632 dihydrochloride anthracyclines treatment. Large scale clinical trials have shown that doxorubicin induced cardiotoxicity is irreversible and dose dependent. Due to advances in basic and clinical cancer research, cancer and malignancies are becoming more manageable, unfortunately the adverse cardiovascular effects of systemic anticancer agents are still a serious concern. Thus it is imperative to understand the cellular and molecular basis of doxorubicin-induced cardiotoxicity with the view to finding therapies that would offer cardioprotection without affecting its anti-tumour effects. Interventions using �� blockers, free radical scavengers, antioxidants and renin-angiotensin system inhibitors have met with limited success due, not only, to side effects but also because of their negative interactions with doxorubicin. While aiming to reduce the cardiotoxic effects of anthracyclines using adjunct therapies, it is imperative to assess the effects in cancer cell line to ascertain the clinical utility of such treatments. Interestingly, recent studies using the phosphodiesterase-5 inhibitors sildenafil or tadalafil have shown promise by showing a reduction in the cardiotoxic effects of doxorubicin without affecting its anti-cancer activity. Cell death pathways activated by doxorubicin treatment usually involve the mitochondria to initiate apoptosis or necrosis. Mitochondrial dynamics are found to play an essential role in cellular function and apoptosis. In order to maintain mitochondrial integrity and efficiency, a constant interplay between mitochondrial fission and fusion is important. Previous studies have demonstrated that upon induction of oxidative stress or ischaemia, dynamin related protein 1, a protein involved in mitochondrial fission, translocates from the cytosol to the mitochondria to execute the mitochondrial division process. This involves hydrolysing GTP, which dysregulates the balance between mitochondrial fusion and fission. Mitochondrial fission leads to cytochrome c release and activation of caspases, which can ultimately lead to cell death. Studies also reported that the dominant negative form of Drp1, DrpK38A, had the ability to inhibit mitochondrial division suggesting a regulatory role for Drp1 in mitochondria-mediated apoptosis.
These responses may contribute to the synergistic effects of silvestrol with cytotoxic agent for HCC cells
Both in vitro and in orthotopic tumor cell xenografts in vivo, and support further development of this agent as a PF-04217903 therapeutic for HCC. Hepatocellular canceris a globally prevalent malignancy for which effective therapies are needed. These studies identify that silvestrol is effective in inhibiting tumor growth in HCC cells both in vitro and in vivo, and thereby provide evidence of a novel therapeutic approach to these cancers. Silvestrol has been shown to inhibit translation, and therapeutic strategies targeting inhibition of protein translation are garnering attention as novel therapeutic agents for diverse human cancers. Thus, our results provide preclinical data that demonstrate the efficacy of targeting translation as a therapeutic strategy for HCC. HCC GDC-0879 frequently arises in the context of chronic hepatic injury resulting in hepatic fibrosis and cirrhosis. The presence of hepatic fibrosis has the potential to impact tumor cell growth and therapeutic impact of anticancer agents. In order to adequately mimic the tumoral microenvironment within which HCC occurs, we developed a disease relevant model of orthotopic HCC tumor cell xenografts in a fibrotic liver to examine the potential in vivo anti-tumor effects of silvestrol. An additional benefit of this model is the ability to examine the effect of anti-cancer therapies under both fibrotic and nonfibrotic conditions, thereby incorporating an assessment of tumor microenvironmental influences on tumor growth as well as anticancer effects. The impact of fibrosis on tumor latency and tumor growth was surprising. While we expected that fibrosis would enhance tumor formation, we were surprised to note that tumor growth was also modulated. Moreover, the differential effects on response to therapy should prompt reevaluation of preclinical data that are derived from in vivo models that do not encompass the fibrotic milieu within which HCC arise. Translation of most mRNAs can be regulated during the ratelimiting stage of initiation. Silvestrol can engage the eukaryotic initiation factor4A and inhibit the initiation of translation by depletion of eIF4A from the eIF4F complex. Importantly, eIF4E is over-expressed in HCC. When eIF4F activity is limiting, mRNAs with a short unstructured 5��-UTR can be translated whereas mRNA with long, G+C rich, highly structured 5��-UTRs are less efficiently translated because efficient ribosome loading is prevented by their complex structure. Thus, the translation of highly structured, malignancy-related mRNAs is sensitive and dependent upon eIF4F for translation. A selective reduction in the translation of mRNAs that contain complex structured 5�� untranslated regions could result from exposure to silvestrol and potentially contribute to its therapeutic efficacy. Over-expression of eIF4E occurs in HCC and in transgenic models has been shown to promote HCC. However, validation of this mechanism as a contributor of the anticancer effects of silvestrol would require selective modulation of eIF4A using other therapeutic strategies, and a systematic analysis of mRNAs that are deregulated by these interventions. Further analysis of selectively translated mRNAs modulated by silvestrol that encode proteins that are engaged in tumor cell survival and growth and tumor cell responses to the tumor microenvironment or to cellular stresses such as chemotherapy may be useful to understand specific pathways involved in HCC progression. Recent studies have suggested  that modulation of protein translation with depletion of survival factors with short half-life can enhance therapeutic responses. Our findings are consistent with these reports by indicating short term effects of silvestrol on Mcl-1 expression and cell apoptosis.
that modulation of protein translation with depletion of survival factors with short half-life can enhance therapeutic responses. Our findings are consistent with these reports by indicating short term effects of silvestrol on Mcl-1 expression and cell apoptosis.