The HDACi SAHA also induced down-regulation of Mcl-1. Furthermore, Mcl-1 protein is a direct cleavage substrate of activated caspase-3. We noted that the amount of Mcl-1 correlated with iuduction of activated caspase-3. Our results demonstrate that SAHA enhances BPR1J-340 inhibition activity in FLT3-ITD + cells and suggests that the enhancement is due to HDACi-induced reduction of FLT3-ITD, STAT5, and Mcl-1. However, the underlying mechanism of enhanced action by combination treatment remains to be further elucidated. The maximum achievable plasma concentration of BPR1J-340 after a single 1.5 mg/kg in rat is more than 272-fold above the IC50 for FLT3-ITD inhibition in biochemical and cellular assays. Even at 24 hour after the single dosing, the plasma levels of BPR1J-340 were close to the IC50 value for inhibition of FLT3ITD. In addition, the high Vss indicated that the distribution of BPR1J-340 into deep tissue compartments, including tumor tissue, is expected. These LY2835219 citations pharmacokinetic properties suggest that BPR1J-340 dosing once a day is sufficient for continuous inhibition of FLT3 activity in rats or mice. To examine whether BPR1J-340 exhibits antitumor activity in vivo, MOLM-13 cells were subcutaneously implanted into nude mice. Our results demonstrated that BPR1J-340 administration resulted in significant tumor regression and tumor shrinkage in this MOLM-13 tumor model. In comparison with sulfonamide BPR1J-97in the same model, BPR1J340 results in a higher CR ratioat a lower dose. These data demonstrated that BPR1J-340 is superior to the sulfonamide compound BPR1J-097 in an in vivo efficacy study. In conclusion, results from this study demonstrate that BPR1J340 exhibits high potency and excellent selectivity against FLT3 kinase, strong suppression of 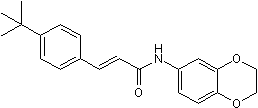 the FLT3-ITD survival signaling pathway, favorable pharmacokinetic properties, and complete tumor regression in a FLT3-ITD xenograft model. These data together support further clinical investigation of PR1J-340 in patients with AML. In addition, the BPR1J-340 potentiated the anti-proliferative activity of the HDAC inhibitor SAHA against human leukemia cells. The combination of SAHA and BPR1J-340 should be a good candidate therapy to develop as a treatment in AML and further investigation in clinical study is warranted. Chronic myeloid leukemiais a myeloproliferative disorder of hematopoietic stem cells GDC-0879 caused by the presence of the BCR-ABL oncogene in the so-called Philadelphia chromosome. The BCR-ABL fusion protein is a constitutively active tyrosine kinase and activator of downstream molecules such as Myc and signal transducer and activator of transcription. BCR-ABL activity promotes the growth of leukemic cells and enhances malignant expansion of hematopoietic stem cells. The clinical outcome for patients with CML is improved by imatinib mesylate. Imatinib was the first ABL tyrosine kinase inhibitorthat was identified to reduce BCRABL kinase activity. In the International Randomized Study of Interferon and STI571trial, imatinib treatment resulted in a high level of cytogenetic response. However, some patients developed resistance to imatinib, which could be attributed to point mutations in the kinase domain of BCR-ABL. These BCR-ABL mutations directly impede contact between the BCRABL protein and imatinib. Recently, second-generation ABL TKIs dasatiniband nilotinibhave been increasingly used for patients resistant to or intolerant of imatinib therapy, and have been approved for front line use in patients with chronic phase CML. However, one point mutation, T315I, located in the gatekeeper region of the ATP-binding site, confers resistance to imatinib, dasatinib, and nilotinib.
the FLT3-ITD survival signaling pathway, favorable pharmacokinetic properties, and complete tumor regression in a FLT3-ITD xenograft model. These data together support further clinical investigation of PR1J-340 in patients with AML. In addition, the BPR1J-340 potentiated the anti-proliferative activity of the HDAC inhibitor SAHA against human leukemia cells. The combination of SAHA and BPR1J-340 should be a good candidate therapy to develop as a treatment in AML and further investigation in clinical study is warranted. Chronic myeloid leukemiais a myeloproliferative disorder of hematopoietic stem cells GDC-0879 caused by the presence of the BCR-ABL oncogene in the so-called Philadelphia chromosome. The BCR-ABL fusion protein is a constitutively active tyrosine kinase and activator of downstream molecules such as Myc and signal transducer and activator of transcription. BCR-ABL activity promotes the growth of leukemic cells and enhances malignant expansion of hematopoietic stem cells. The clinical outcome for patients with CML is improved by imatinib mesylate. Imatinib was the first ABL tyrosine kinase inhibitorthat was identified to reduce BCRABL kinase activity. In the International Randomized Study of Interferon and STI571trial, imatinib treatment resulted in a high level of cytogenetic response. However, some patients developed resistance to imatinib, which could be attributed to point mutations in the kinase domain of BCR-ABL. These BCR-ABL mutations directly impede contact between the BCRABL protein and imatinib. Recently, second-generation ABL TKIs dasatiniband nilotinibhave been increasingly used for patients resistant to or intolerant of imatinib therapy, and have been approved for front line use in patients with chronic phase CML. However, one point mutation, T315I, located in the gatekeeper region of the ATP-binding site, confers resistance to imatinib, dasatinib, and nilotinib.
Author: screening library
A promising novel strategy in anti-angiogenic therapy specifically protects cells against proteasome inhibitor-induced apoptosis
Since proteasome inhibitors down-regulate FoxM1 and FoxM1 overexpression protects against cell death mediated by proteasome inhibitors, suppression of FoxM1 may be required for the anticancer activity of these drugs. Further experiments are needed to clarify the mechanisms of FoxM1 down-regulation by proteasome inhibitors and the importance of FoxM1 suppression in the activity of proteasome inhibitors as anticancer drugs. Angiogenesis, the sprouting of new vessels from the existing vasculature, mainly occurs during embryonic development and growth. In the adult it is restricted to distinct physiological processes, e.g. wound healing, by a balance of pro- and antiangiogenic factors. Unregulated angiogenesis is one of the hallmarks of 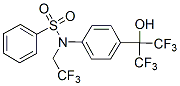 cancer. Tumor growth is highly dependent on proper supply with oxygen and nutrients and removal of metabolic waste. Therefore, angiogenesis is crucial for tumor survival and proliferation, and tumor size remains limited unless the tumor switches to an angiogenic phenotype. The intent to stop tumor growth and finally starve the tumor by disrupting angiogenic signaling has led to the development of anti-angiogenic drugs for anticancer therapy. Agents addressing vascular endothelial growth factorinduced angiogenesis have already been successfully introduced into tumor therapy. However, in clinical use it has become apparent that antiangiogenic tumor therapy is more challenging than expected: Many tumors are refractory to VEGF-blockade or become resistant during treatment. This evasive resistancecan be caused by a shift to alternative angiogenic signaling pathways due to a pre-existing multiplicity of redundant pro-angiogenic signals. Therefore novel targets in angiogenesis need to be identified and characterized as a basis for future INCB28060 therapeutic concepts. Cdk5 has been discovered as a neuronal cdc2-like serine/ threonine kinasein 1992. Despite its high sequence homology with the mitotic Cdk1, Cdk5 is not involved in cell cycle control and unique among the Cdks in its regulation and function. On the cellular level, Cdk5 is well-described in neurons as the key hub in the dynamic network of trafficking and transport, integrating signals in cytoskeletal dynamics during neuronal migration, in synaptic plasticity and synaptic vesicle endo- and exocytosis, cell adhesion and axon guidance, neuromuscular development and pain signaling. Although Cdk5 expression and activity is highest in the central nervous system, Cdk5 is also expressed in various tissues, and an increasing body of research uncovers extraneuronal functions of Cdk5, where it is involved in the regulation of migration, cell death and survival, glucose metabolism and inflammation. -roscovitine or CYC-202/seliciclib �C in the following referred to as roscovitine �C belongs to the class of 2,6,9-trisubstituted purines. It is one of the best-known Cdk inhibitors, and is currently tested in several phase I and phase II clinical trials for tumor treatment. Roscovitine inhibits mainly Cdk1, Cdk2, Cdk5, Cdk7 and Cdk9 and exerts anti-mitotic and Foretinib pro-apoptotic effects in a wide range of tumor cells. Cell-cycle independent actions of roscovitine mainly derive from Cdk5 inhibition and include anti-angiogenicand anti-inflammatoryeffects, inhibition of cell migration and motilityand modulation of glucose metabolism. Anti-angiogenic actions of Cdk inhibitors have been observed in vitro and in vivo. Recently, we have demonstrated that the anti-angiogenic effect of roscovitine most likely results from impaired endothelial cell migration. The effect on migration was traced down to Cdk5 inhibition which led to Rac1 inactivation and lamellipodia disruption.
cancer. Tumor growth is highly dependent on proper supply with oxygen and nutrients and removal of metabolic waste. Therefore, angiogenesis is crucial for tumor survival and proliferation, and tumor size remains limited unless the tumor switches to an angiogenic phenotype. The intent to stop tumor growth and finally starve the tumor by disrupting angiogenic signaling has led to the development of anti-angiogenic drugs for anticancer therapy. Agents addressing vascular endothelial growth factorinduced angiogenesis have already been successfully introduced into tumor therapy. However, in clinical use it has become apparent that antiangiogenic tumor therapy is more challenging than expected: Many tumors are refractory to VEGF-blockade or become resistant during treatment. This evasive resistancecan be caused by a shift to alternative angiogenic signaling pathways due to a pre-existing multiplicity of redundant pro-angiogenic signals. Therefore novel targets in angiogenesis need to be identified and characterized as a basis for future INCB28060 therapeutic concepts. Cdk5 has been discovered as a neuronal cdc2-like serine/ threonine kinasein 1992. Despite its high sequence homology with the mitotic Cdk1, Cdk5 is not involved in cell cycle control and unique among the Cdks in its regulation and function. On the cellular level, Cdk5 is well-described in neurons as the key hub in the dynamic network of trafficking and transport, integrating signals in cytoskeletal dynamics during neuronal migration, in synaptic plasticity and synaptic vesicle endo- and exocytosis, cell adhesion and axon guidance, neuromuscular development and pain signaling. Although Cdk5 expression and activity is highest in the central nervous system, Cdk5 is also expressed in various tissues, and an increasing body of research uncovers extraneuronal functions of Cdk5, where it is involved in the regulation of migration, cell death and survival, glucose metabolism and inflammation. -roscovitine or CYC-202/seliciclib �C in the following referred to as roscovitine �C belongs to the class of 2,6,9-trisubstituted purines. It is one of the best-known Cdk inhibitors, and is currently tested in several phase I and phase II clinical trials for tumor treatment. Roscovitine inhibits mainly Cdk1, Cdk2, Cdk5, Cdk7 and Cdk9 and exerts anti-mitotic and Foretinib pro-apoptotic effects in a wide range of tumor cells. Cell-cycle independent actions of roscovitine mainly derive from Cdk5 inhibition and include anti-angiogenicand anti-inflammatoryeffects, inhibition of cell migration and motilityand modulation of glucose metabolism. Anti-angiogenic actions of Cdk inhibitors have been observed in vitro and in vivo. Recently, we have demonstrated that the anti-angiogenic effect of roscovitine most likely results from impaired endothelial cell migration. The effect on migration was traced down to Cdk5 inhibition which led to Rac1 inactivation and lamellipodia disruption.
Binding interactions of the sulfonamide MurD inhibitors and the observed dynamic behavior of ligand-MurD
Complexes are in agreement with the crucial NMR experimental findings about the binding mode of these inhibitors.The rigid D-Glu mimetics of second generation sulfonamide inhibitors form stable electrostatic interactions with the D-Glu-binding site, which is supported by their large effects on the CPSs of methyl ICG-001 groups near the D-Glu-binding site.The C6 arylalkyloxy substituents are stabilized in the uracil-binding pocket with a number of stable electrostatic and hydrophobic interactions. This is in agreement with their pronounced effects on the CSPs of methyl groups near the uracil binding site.The C6 alkyloxy substituents are flexible in the uracil-binding site, forming weaker hydrophobic interactions; the CSPs of methyl groups near the uracil binding site are significantly lower.The naphthalene ring rotations are supported by the NOE patterns of bound ligands.The type of substitution of rigid D-Glu mimetic significantly effects the electrostatic interactions of the sulfonamide group with the central domain. This is supported by the pronounced effects of 6b on the CPSs belonging to the central domain residues. MurD conformational changes have to date been given insufficient attention in the process of MurD inhibitor optimization. MD simulations show the complex dynamic behavior of these MurD�Cinhibitor complexes, where the interactions are affected both by movements of the protein domains and by the flexibility of the ligand. The differing degrees of conformational flexibility of the ligands were also predicted on the basis of the NOE patterns. The sulfonamide inhibitors studied span from the C-terminal domain to the N-terminal domain and also interact with the central domain. The distances between the C-terminal and Nterminal domains fluctuate. Therefore, the bound ligands are exposed to Niraparib stretching forces that tend to pull either the D-Glu mimetic part or the C6 substituent out of the binding site. Stronger interactions in one domain tend to weaken the interactions in the other domains. This needs to be considered in the optimization of these sulfonamide inhibitors through the design of new compounds that have improved interactions not only with one but with both the C-terminal and N-terminal domains of MurD. Our data also suggest that inhibitors that can span from the C-terminal domain to the N-terminal domain should not be highly rigid to allow them to adapt to the conformational changes of the MurD protein. Such compounds might also benefit from having slightly longer linkers between the naphthalene rings and the aromatic rings of the C6 substituent. These data represent upgraded knowledge that will now be useful for the rational structure-based design of new improved MurD inhibitors. STD ligand epitope mappingwas performed with an 8389 Hz spectral width, with 16384 data points, a saturation time of 350 ms, a relaxation delay of 11.35s, and 3000 to 8000 scans. The spectra were recorded at a protein/ligand ratio of 1:100. Selective saturation was achieved by a train of 50 ms long Gaussshaped pulses, separated by 1 ms delays. Water was suppressed via excitation sculpting. The on-resonance selective saturation of MurD was applied at 0.21 ppm. The off-resonance irradiation was applied at 30 ppm for the reference spectrum. Subtraction of the on-resonance and off-resonance spectra was performed internally via phase cycling. The transferred NOESYspectra were acquired at a protein/ligand ratio of 1:45,  with an 8389 Hz spectral width, with 4096 data points in t2, 32�C48 scans, 256�C356 complex points in t1, a mixing time of 250 ms, and a relaxation delay of 1.5 s. The residual water signal was suppressed using excitation sculpting, and adiabatic pulseswere applied for suppression.
with an 8389 Hz spectral width, with 4096 data points in t2, 32�C48 scans, 256�C356 complex points in t1, a mixing time of 250 ms, and a relaxation delay of 1.5 s. The residual water signal was suppressed using excitation sculpting, and adiabatic pulseswere applied for suppression.
The lesson learned from nearly a century of experience with antibiotics is that pathogens present a moving target
To understand the robustness of combination inhibitor therapies across diverse bacterial species, we test each strategy against a number of biochemical and transcriptional variants of the experimentally validated QS model. We find that a combination of LuxI and LuxR non-competitive inhibitors act multiplicatively to inhibit virulence for a broad range of QS systems. In contrast, we find that LuxR competitive inhibitors act antagonistically with LuxI inhibitors, due to the weak activation of LuxR; in some conditions this can actually increase virulence. Both these results are somewhat surprising, and seem to arise due to the global structure of QS systems. Combination therapies must therefore be used with care, only once the most relevant drug combinations and molecular targets have been identified for each pathogenic species and infection context. QS inhibitors are promising alternatives to antibiotics, but there are still many steps on the path to their widespread use. It has been argued that pathogens targeted with QS inhibitors would be under weaker selective pressure to develop resistance, compared to the MLN4924 pressures induced by antibiotics. However, the reality is more complex: in an infection context, individuals resistant to QS inhibition have a major advantage, and tend to be selected. Combination drug therapies that target multiple molecules simultaneously would lower the rate at which such resistant  individuals spontaneously arose. This motivated us to ask which QS targets would respond best to simultaneous inhibition. QS being implemented by a non-linear feedback system, the answer to such a question is far from obvious: it will vary from one pathogen to another, depending on the underlying feedback Fingolimod topology and biochemical parameter values. However, our analysis does produce some robust results. We find that a combination of LuxI inhibitors and LuxR noncompetitive inhibitors has the greatest capacity to suppress virulence, across a wide range of parameters. This strategy should be considered as the default: it can be applied without detailed knowledge of the pathogen��s QS system; moreover, since it targets two distinct molecules, the likelihood of spontaneous resistance is reduced. In contrast, LuxR competitive inhibitors should be used with care. These molecules tend to be AHL analogues with some weak capacity to activate LuxR. Though this capacity is much less than that of AHL itself when measured per molecule, the overall effect depends sensitively on AHL levels at the site of infection. Since physiological AHL concentrations tend to be low, competitive inhibitors in the form of AHL analogues can paradoxically increase virulence gene expression. Recently, structure-function studies have been used to design AHL analogues that completely block LuxR-DNA binding. Such studies show that LuxR competitive inhibitors might yet find use. New experiments can help improve the design of anti-virulence therapies at three levels. First: Any mathematical model such as ours is limited by the accuracy of the equations used, and of the parameter values they contain. Careful biochemical measurements can improve the predictive power of these models. Second: no mathematical model can account for the complications of realworld therapy. The true test of any strategy can only come from experimental studies on animal models, and ultimately from clinical trials. Third: our results should be understood in the context of a wider range of strategies, including the use of QS inhibitors in combination with antibiotics, as well as the stimulation of the host immune system. Research on pathogen biology will add to this list, revealing new and unexpected strategies.
individuals spontaneously arose. This motivated us to ask which QS targets would respond best to simultaneous inhibition. QS being implemented by a non-linear feedback system, the answer to such a question is far from obvious: it will vary from one pathogen to another, depending on the underlying feedback Fingolimod topology and biochemical parameter values. However, our analysis does produce some robust results. We find that a combination of LuxI inhibitors and LuxR noncompetitive inhibitors has the greatest capacity to suppress virulence, across a wide range of parameters. This strategy should be considered as the default: it can be applied without detailed knowledge of the pathogen��s QS system; moreover, since it targets two distinct molecules, the likelihood of spontaneous resistance is reduced. In contrast, LuxR competitive inhibitors should be used with care. These molecules tend to be AHL analogues with some weak capacity to activate LuxR. Though this capacity is much less than that of AHL itself when measured per molecule, the overall effect depends sensitively on AHL levels at the site of infection. Since physiological AHL concentrations tend to be low, competitive inhibitors in the form of AHL analogues can paradoxically increase virulence gene expression. Recently, structure-function studies have been used to design AHL analogues that completely block LuxR-DNA binding. Such studies show that LuxR competitive inhibitors might yet find use. New experiments can help improve the design of anti-virulence therapies at three levels. First: Any mathematical model such as ours is limited by the accuracy of the equations used, and of the parameter values they contain. Careful biochemical measurements can improve the predictive power of these models. Second: no mathematical model can account for the complications of realworld therapy. The true test of any strategy can only come from experimental studies on animal models, and ultimately from clinical trials. Third: our results should be understood in the context of a wider range of strategies, including the use of QS inhibitors in combination with antibiotics, as well as the stimulation of the host immune system. Research on pathogen biology will add to this list, revealing new and unexpected strategies.
Moieties protrude into the NAM binding site and mimic the natural substrate to form covalent adducts with PRPP
NAMPT mutations that confer resistance to GMX1778, APO866 or TP201565, a structural analog of GMX1778 have been mapped to G217R, H191R, D93del, and Q388R. Based on the wild-type LY294002 enzyme structure, residues G217R and H191R appeared to protrude into the inhibitor-binding pocket, while D93 and Q388 are located on the dimer interface. In this work, we identify and characterize six mutations in NAMPT that confer resistance to a novel small molecule inhibitor of NAMPT, GNE-618. These include G217R, D93del as well as 4 previously unreported mutations. Furthermore, we determine the crystal structures of six NAMPT mutants in the apo form and in complex with various inhibitors and present a definitive model to explain the differential effects of the mutations on various structural classes of NAMPT inhibitors. We identified and characterized a variety of NAMPT protein mutations mediating resistance against the bi-aryl sulfone inhibitors, exemplified by GNE-618. The identification of resistance mutations in S165 is unexpected given its distance from the inhibitor-binding site. However, expression of S165F or S165Y mutant NAMPT proteins in a naive cell line resulted in decreased sensitivity to GNE-618, indicating that these mutations are sufficient to cause resistance to this NAMPT inhibitor. Furthermore, xenografts  derived from cells harboring the S165Y mutation in NAMPT are resistant to GNE-618 at doses that are efficacious in the parental line, suggesting that this is relevant in vivo. Structural analyses of S165 NAMPT mutant proteins establish the critical role of the 380GRS in NAMPT catalysis. Crystal structures revealed a previously underappreciated conformational flexibility in this secondary structure element that can be exploited by resistance mutations through an allosteric mechanism. Our finding regarding the S165F/Y mechanism of action provides an explanation regarding another mutant, Q388R, previously reported to cause resistance to GMX1778. Q388 is located at the C-terminal end of the 380GRS, and is normally buried under the protein surface. It is unlikely for an arginine residue to settle in the same space and maintain the wild-type ALK5 Inhibitor II conformation of the helix. Given the flexibility associated with the 380GRS revealed by our analysis, we propose that R388 is likely to drive it into an alternative conformation that can negatively impact NAMPT inhibitor potency. In conclusion, we present a systematic approach of identifying resistant mutations and elucidating their mechanism of action. Our work provides the most comprehensive to-date structural analysis of resistance mutations to NAMPT inhibitors, important for future rational drug design of more effective inhibitors against this target and similar classes of enzymes and for better understanding of the catalytic mechanism for this important metabolic enzyme. Virus infection triggers the cellular interferonresponse to produce Type 1 IFN’s alpha and beta. Secreted IFNa/b can stimulate the JAK-STAT pathway in an autocrine or paracrine manner to activate hundreds of IFN-stimulated genes, many of which have antiviral activities that elicit an antiviral state. Although the IFN system constitutes a powerful antiviral response, it rarely works to full capacity because virusencoded IFN antagonists circumvent it. Manipulation of a virus’s capacity to circumvent the IFN response enables both basic research and various practical applications. For example, genetic engineering has facilitated rational design of live-attenuated vaccines, where a common approach is to disable a virus’s IFN antagonist thereby restricting its ability to circumvent the IFN response.
derived from cells harboring the S165Y mutation in NAMPT are resistant to GNE-618 at doses that are efficacious in the parental line, suggesting that this is relevant in vivo. Structural analyses of S165 NAMPT mutant proteins establish the critical role of the 380GRS in NAMPT catalysis. Crystal structures revealed a previously underappreciated conformational flexibility in this secondary structure element that can be exploited by resistance mutations through an allosteric mechanism. Our finding regarding the S165F/Y mechanism of action provides an explanation regarding another mutant, Q388R, previously reported to cause resistance to GMX1778. Q388 is located at the C-terminal end of the 380GRS, and is normally buried under the protein surface. It is unlikely for an arginine residue to settle in the same space and maintain the wild-type ALK5 Inhibitor II conformation of the helix. Given the flexibility associated with the 380GRS revealed by our analysis, we propose that R388 is likely to drive it into an alternative conformation that can negatively impact NAMPT inhibitor potency. In conclusion, we present a systematic approach of identifying resistant mutations and elucidating their mechanism of action. Our work provides the most comprehensive to-date structural analysis of resistance mutations to NAMPT inhibitors, important for future rational drug design of more effective inhibitors against this target and similar classes of enzymes and for better understanding of the catalytic mechanism for this important metabolic enzyme. Virus infection triggers the cellular interferonresponse to produce Type 1 IFN’s alpha and beta. Secreted IFNa/b can stimulate the JAK-STAT pathway in an autocrine or paracrine manner to activate hundreds of IFN-stimulated genes, many of which have antiviral activities that elicit an antiviral state. Although the IFN system constitutes a powerful antiviral response, it rarely works to full capacity because virusencoded IFN antagonists circumvent it. Manipulation of a virus’s capacity to circumvent the IFN response enables both basic research and various practical applications. For example, genetic engineering has facilitated rational design of live-attenuated vaccines, where a common approach is to disable a virus’s IFN antagonist thereby restricting its ability to circumvent the IFN response.