As such, the predicted complete release of IQP-0410 from the film is calculated to occur after 19.5 days from application and 50% release is calculated to occur after 9.75 days of use. The reduction in drug flux is to be expected and can be directly attributed to the difference in diffusion of IQP-0410 through epidermal tissue and a thin membrane. Most importantly, despite initial predictions that a drug with the physicochemical properties of IQP-0410 would remain in the stratum corneum, IQP-0410 was successfully released and permeated through the full thickness epidermal tissue to be collected in the basal media suggesting the potential viability of controlled zero-order delivery of IQP-0410 through the skin. While the in vitro release of IQP-0410 into dissolution media suggested an increased rate from films stored under accelerated storage conditions, this was not observed in the ex vivo permeability studies. Masitinib 790299-79-5 Transdermal drug delivery has two parts for overall drug delivery: release from formulation to the epidermis, and permeation through the skin to the underlying blood vessels. Therefore any increases in drug release rate from the film formulation will be mitigated by the diffusion of the drug through the tissue. The passive diffusion across the skin, the basis of transdermal drug 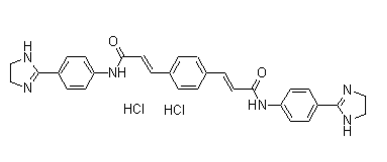 delivery, will be the limiting factor in the drug delivery and permeability. Only through external interventions to increase skin permeability, such as chemical solubility enhancers, thermal ablation, microneedles, and iontophoresis, will increased drug release from the formulations effect overall transdermal film drug delivery. To evaluate the anti-HIV efficacy of the delivered IQP-0410 through the epidermal tissue, in vitro assays in CEM-SS cells and PBMC��s against HIV-1 were performed with the collected IQP-0410 from the basal media. The concentration of IQP-0410 detected in the basal media was defined as the amount of IQP-0410 potentially bioavailable systemically and then was used to evaluate the in vitro anti-HIV efficacy and cellular toxicity performed in CEM-SS cells and PBMCs. In the basal media, IQP-0410 was collected each day, respectively, resulting in an average EC50 value in CEM-SS cells and in PBMCs over a three day application. Therefore, in conjunction with the drug recovery evaluations, we are assured that this transdermal film formulation has neither negative physicochemical nor biological effects on IQP-0410. Ultimately it is important to evaluate whether the transdermal film is capable of delivering appropriate amounts of drug into the system. Ex vivo release studies performed over three days determined that the concentration of delivered IQP-0410 after 24 hours is 5,000 fold greater than in vitro EC95 values. It is understood that the ex vivo concentrations of permeated IQP-0410 are not completely representative of potential systemic in vivo behavior; however, it can be concluded that efficacious concentrations of IQP-0410 are passing through the skin. In addition, previous product profile studies of IQP-0410 have shown that in vivo oral administration in mice results in 24% oral bioavailability with a half-life of 5.37 hours and in the presence of liver microsomes, the expected half-life in humans is 15.7 minutes. Therefore, it will be critical to avoid first pass metabolism in the liver commonly observed in oral administration to maintain therapeutic concentrations of IQP-0410 in the blood plasma by controlled delivery through the skin. One the greatest disadvantage to transdermal delivery is the possibility that a local irritation will develop at the site of administration. Irritation can be caused by the drug itself, the adhesive, or other excipients in the formulation. When applied to the skin tissue for 24 and 72 hours, the IQP-0410 transdermal films had a tissue viability of 121 �� 19.8% after a 24 hour exposure and 91.2 �� 9.71% tissue viability after 72 hours. The in vitro toxicity results from the tissue do offer a correlation in predicting clinical in vivo skin irritation. So, from the MTT TWS119 in vivo analysis of the IQP-0410 transdermal film, an in vitro ET-50 >24 hours is equivalent to the non-irritancy of 10% Tween-20. Therefore, it is expected that the IQP-0410 film formulation will result in no adverse skin irritation when administered.
delivery, will be the limiting factor in the drug delivery and permeability. Only through external interventions to increase skin permeability, such as chemical solubility enhancers, thermal ablation, microneedles, and iontophoresis, will increased drug release from the formulations effect overall transdermal film drug delivery. To evaluate the anti-HIV efficacy of the delivered IQP-0410 through the epidermal tissue, in vitro assays in CEM-SS cells and PBMC��s against HIV-1 were performed with the collected IQP-0410 from the basal media. The concentration of IQP-0410 detected in the basal media was defined as the amount of IQP-0410 potentially bioavailable systemically and then was used to evaluate the in vitro anti-HIV efficacy and cellular toxicity performed in CEM-SS cells and PBMCs. In the basal media, IQP-0410 was collected each day, respectively, resulting in an average EC50 value in CEM-SS cells and in PBMCs over a three day application. Therefore, in conjunction with the drug recovery evaluations, we are assured that this transdermal film formulation has neither negative physicochemical nor biological effects on IQP-0410. Ultimately it is important to evaluate whether the transdermal film is capable of delivering appropriate amounts of drug into the system. Ex vivo release studies performed over three days determined that the concentration of delivered IQP-0410 after 24 hours is 5,000 fold greater than in vitro EC95 values. It is understood that the ex vivo concentrations of permeated IQP-0410 are not completely representative of potential systemic in vivo behavior; however, it can be concluded that efficacious concentrations of IQP-0410 are passing through the skin. In addition, previous product profile studies of IQP-0410 have shown that in vivo oral administration in mice results in 24% oral bioavailability with a half-life of 5.37 hours and in the presence of liver microsomes, the expected half-life in humans is 15.7 minutes. Therefore, it will be critical to avoid first pass metabolism in the liver commonly observed in oral administration to maintain therapeutic concentrations of IQP-0410 in the blood plasma by controlled delivery through the skin. One the greatest disadvantage to transdermal delivery is the possibility that a local irritation will develop at the site of administration. Irritation can be caused by the drug itself, the adhesive, or other excipients in the formulation. When applied to the skin tissue for 24 and 72 hours, the IQP-0410 transdermal films had a tissue viability of 121 �� 19.8% after a 24 hour exposure and 91.2 �� 9.71% tissue viability after 72 hours. The in vitro toxicity results from the tissue do offer a correlation in predicting clinical in vivo skin irritation. So, from the MTT TWS119 in vivo analysis of the IQP-0410 transdermal film, an in vitro ET-50 >24 hours is equivalent to the non-irritancy of 10% Tween-20. Therefore, it is expected that the IQP-0410 film formulation will result in no adverse skin irritation when administered.