The SVLP signal faded at later time points, but the loss of signal was still gradual over a number of days. This kinetics of uptake and probable processing is consistent with the report that DC process antigen slowly. However, it was still unclear if the slow kinetics were related to intracellular processing or simply the rate of internalisation. Primary DC and monocyte-derived DC are inefficient at functional DNA transfection, possibly due to the impermeability and resistance of their nuclear membranes. It was considered inappropriate to employ cell lines, because the aim of the work was to characterize how primary DC handled a complex antigen with proven in vivo efficacy. Accordingly, the intracellular compartments with which the SVLP associated were analysed using ligands and antibodies for their identification. SVLP interacted with lipid rafts on the DC surface. This was concluded due to their patchy association with the cell surface on ice, accumulation at the leading edge of the cell on shifting to 39uC, and co-localisation with CTB, which is known to interact with lipid rafts. Co-localisation of the SVLP with the CD9 marker associated with lipid rafts of human monocytes, and sensitivity to MBCD treatment, known to interfere with cholesterol restoration, confirmed that SVLP endocytosis by DC was dominated by lipid raft-dependent processes. These were caveolin-independent, because there was no co-localisation of SVLPcontaining structures with caveolin-1. This kinetics followed that of DQ-Ova, a self-quenched entity employed for detecting endosomal-processing events, the high degree of overlap indicating similar processing pathways. This confirmed slow enzymatic processing, but also indicated that the SVLP-containing vesicles not associated with EEA-1 had probably not interacted with early endosomes. Although the Benzethonium Chloride results were indicating an important role for lipidraft mediated macropinocytosis, there was evidence of an early involvement for a clathrin-dependent route. Nevertheless, most SVLP were not associated with clathrin + structures, and the latter were only visible during the first 10 min. This implies that clathrin-dependent endocytosis is of minor relevance to the processing of SVLP for antigen presentation; indeed, the early clathrin-dependent uptake may be leading the SVLP into a rapid degradative process, rather than the slower and more progressive antigen-processing route. The results with transferrin would also argue in this direction. While transferrin was continually observed translocating from the periphery, the SVLP tended to remain more peripheral than the transferrin. Some peripheral vesicles did contain both SVLP and transferrin, but this was rather late �C at the 60-min time point. This may reflect diversion of some SVLPcontaining vesicles into sorting endosomes. Although such structures were in the minority, such a possibility relates to the sorting of antigen for delivery to B-lymphocytes. The importance of macropinocytosis in SVLP internalisation by DC was confirmed using high molecular weight dextran, which should remain blocked within macropinosomes. Allowing dextran to accumulate in macropinosomes, SVLP-containing vesicles entering at the leading edge of the DC were not associated with dextran, confirming their early endocytic stage. By 20 min at 39uC, most of the SVLP Mepiroxol co-localised with the dextran+ vesicles, strongly indicative of SVLP accumulation in macropinosomes. This would certainly relate to the importance of macropinocytosis for the entry of viruses into DC. Use of maturing DC for their reduced macropinocytic activity further confirmed the importance of macropinocytosis during SVLP uptake. Mature DC were inferior to immature 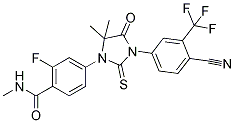 DC for endocytosis of SVLP; in contrast to high molecular weight dextran, the uptake of which was abrogated in mature DC, there was a residual uptake of SVLP.
DC for endocytosis of SVLP; in contrast to high molecular weight dextran, the uptake of which was abrogated in mature DC, there was a residual uptake of SVLP.