With C/EBPb gene was shown to lead to an increase in the expression of Gsa subunit, cAMP accumulation, enhanced mitochondrial biogenesis and Ucp1 expression in WAT. WAT in mice with an overexpression of FoxC2 acquired certain BAT-like properties with increased expression levels of PGC1, Ucp1 and cAMP pathway proteins such as b3-adrenoceptor and the protein kinase A alpha regulatory subunit1. Similarly, in mice lacking negative regulators for BAT differentiation like p107, Rb and RIP140, there is a uniform replacement of WAT with BAT and elevated levels of PGC1a and Ucp1. Although many factors and regulatory pathways have been shown to play important roles in maintaining WAT identity, upstream signals or factors that determine the fate of BAT vs WAT and those that initiate the conversion of WAT to BAT remain unclear. Cide proteins including Cidea, Cideb and Fat specific protein 27. Our previous work demonstrated that Cidea is expressed at high levels in BAT, whereas Cideb is expressed at high levels in liver. Mice with deficiency in both Cidea and Cideb have higher energy expenditure, enhanced insulin 20S-Notoginsenoside-R2 sensitivity and are resistance to high-fat-diet -induced obesity and diabetes, suggesting a universal role of Cide proteins in the regulation of energy homeostasis. Fsp27 was originally identified in differentiated TA1 adipocytes and its expression is regulated by C/ EBP transcription factor. Fsp27 mRNA is detected at high levels in WAT and moderate levels in BAT. Lower levels of Fsp27 mRNA are present in skeletal muscle and lung. In human, it is also detected in heart and many other tissues. Furthermore, Fsp27 protein is detected in the lipid droplet enriched fraction and over-expressed Fsp27 protein was shown to be targeted to 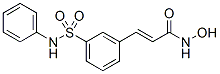 lipid droplets and promotes TAG storage. Recently, Fsp27 was also found to be a direct mediator of PPARc-dependent hepatic steatosis. However, its physiological role in regulating adipocyte function and the development of obesity is largely unknown. Here, we observed that Fsp27 protein was detected in both BAT and WAT, was dramatically up-regulated in the WAT and liver of ob/ob mice suggesting that Fsp27 expression is positively correlated with the development of obesity. Fsp27 deficiency results in dramatically reduced WAT depot and the acquisition of a brown fat-like morphology in these WAT, typified by the appearance of smaller lipid droplets, increased mitochondrial size and their activity. Furthermore, both Fsp27 deficient and Fsp27/leptin double deficient mice display improved insulin sensitivity and a lean phenotype. The incidence of cancer increases with age, suggesting that physiological changes associated with aging contribute to carcinogenesis. Cellular proliferation over the course of a lifetime coincides with a progressive loss of DNA from the ends of chromosomes, or telomeres, which culminates in a cessation of cell division known as replicative senescence. In yeast, replicative senescence appears to be concurrent with telomere Albaspidin-AA dysfunction and crisis, a precipitous loss of cell viability. However, in vertebrates the subsequent loss of the tumor suppressors p53 or Rb in a rare subset of cells is required to escape replicative senescence and undergo the further telomere shortening that leads to telomere dysfunction and crisis. Such critical shortening of the telomeres in the cells of late-generation mice unable to replicate their telomeres can lead to a loss of telomere function that potentiates the development of epithelial cancers. These tumors display aneuploidy and non-reciprocal translocations that are also characteristic of the epithelial tumors of adult humans.
lipid droplets and promotes TAG storage. Recently, Fsp27 was also found to be a direct mediator of PPARc-dependent hepatic steatosis. However, its physiological role in regulating adipocyte function and the development of obesity is largely unknown. Here, we observed that Fsp27 protein was detected in both BAT and WAT, was dramatically up-regulated in the WAT and liver of ob/ob mice suggesting that Fsp27 expression is positively correlated with the development of obesity. Fsp27 deficiency results in dramatically reduced WAT depot and the acquisition of a brown fat-like morphology in these WAT, typified by the appearance of smaller lipid droplets, increased mitochondrial size and their activity. Furthermore, both Fsp27 deficient and Fsp27/leptin double deficient mice display improved insulin sensitivity and a lean phenotype. The incidence of cancer increases with age, suggesting that physiological changes associated with aging contribute to carcinogenesis. Cellular proliferation over the course of a lifetime coincides with a progressive loss of DNA from the ends of chromosomes, or telomeres, which culminates in a cessation of cell division known as replicative senescence. In yeast, replicative senescence appears to be concurrent with telomere Albaspidin-AA dysfunction and crisis, a precipitous loss of cell viability. However, in vertebrates the subsequent loss of the tumor suppressors p53 or Rb in a rare subset of cells is required to escape replicative senescence and undergo the further telomere shortening that leads to telomere dysfunction and crisis. Such critical shortening of the telomeres in the cells of late-generation mice unable to replicate their telomeres can lead to a loss of telomere function that potentiates the development of epithelial cancers. These tumors display aneuploidy and non-reciprocal translocations that are also characteristic of the epithelial tumors of adult humans.