Six months after implantation, the weight average molecular weight of the in vivo and in vitro stents had respectively decreased to 61.8% and 68.5% of the initial molecular weight. As it can be seen in Figure 6, the decrease in the number average molecular weight of the stents over the 0 to 6 months after implantation in the in vivo and in vitro models was consistent. The molecular weight of the bioabsorbable material decreased along the process of degradation, but the loss in mass or the fracture of the material occurred only when the decrease in the molecular weight attained a certain threshold value. The degradation would start impacting the supporting performance of the stent only when the threshold value was attained. However, since this study only explored the early stages of degradation behavior; even though certain parts of the stent underwent accelerated degradation, the weight-averaged molecular weight and overall supporting performance of the stent were not significantly affected. The outer diameters of the stent from the in vitro and in vivo models are illustrated in Figure 7. The stent Ginsenoside-F4 diameter difference of in vivo and in vitro tests is mainly because of different measurement methods and the artery size variations between the in vitro and in vivo tests. Up to certain extent, the changes in the diameter of the stents reflected whether the stents had sufficient radial support and strength. The size of the stent did not reduce significantly over the degradation time, proving that when the stent was implanted into the blood vessels, it was able to function very well to support the blood vessels. It was also found that the change trend of the stent diameter was consistent in both in vitro and in vivo experiments. Hence, it was observed that during the in vivo and in vitro degradation process, the outer diameter of 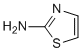 the implanted stent did not undergo significant changes over time, meaning that the stents were able to sustain appropriate radial strength. The initial properties of the bioabsorbable material prior to degradation were used to simulate the finite element analysis of the un-degraded stent. In the first step, the outer diameter of the stent was reduced to 1.5 mm by using a crimping tool, hence simulating the crimping analysis. From Figure 8, it can be seen that stress and strain were concentrated in the central region of the reinforcing unit of the stent. In the second step, the Folic acid elastic recoil of the stent after crimping was simulated by removing the crimping tool; As it can be seen from the stress distribution, the stress in the stent decreased from 161 MPa to 102.4 MPa after the elastic recoil. Eventually, after the crimping analysis, the stent was expanded. Figure 8 illustrates the stress and strain distributions of the stent when it was fully expanded and recoiled after expansion. As it can be seen from Figure 8, when the stent was fully expanded, the stress and strain were mainly concentrated at the central location, nearing the surface area of the inner curvature of the reinforced ring, where the tensile stress had reached 172.3 MPa with the strain value of 120.7%.The Goodman diagram was used to illustrate the fatigue state of the stent while the distribution of the alternating stress points and average stress points are illustrated as in Figure 8. The finite element model simulation of the degradation process was performed on the stent at an equilibrium state after the fatigue analysis, where the changes in the degradation state of the stent and the distribution of the degradation degree were analyzed. Figure 9 illustrates the changes in the degradation degree and the comparison of the degradation degree vs. time at different positions of the stent over 30 days of the degradation process.
the implanted stent did not undergo significant changes over time, meaning that the stents were able to sustain appropriate radial strength. The initial properties of the bioabsorbable material prior to degradation were used to simulate the finite element analysis of the un-degraded stent. In the first step, the outer diameter of the stent was reduced to 1.5 mm by using a crimping tool, hence simulating the crimping analysis. From Figure 8, it can be seen that stress and strain were concentrated in the central region of the reinforcing unit of the stent. In the second step, the Folic acid elastic recoil of the stent after crimping was simulated by removing the crimping tool; As it can be seen from the stress distribution, the stress in the stent decreased from 161 MPa to 102.4 MPa after the elastic recoil. Eventually, after the crimping analysis, the stent was expanded. Figure 8 illustrates the stress and strain distributions of the stent when it was fully expanded and recoiled after expansion. As it can be seen from Figure 8, when the stent was fully expanded, the stress and strain were mainly concentrated at the central location, nearing the surface area of the inner curvature of the reinforced ring, where the tensile stress had reached 172.3 MPa with the strain value of 120.7%.The Goodman diagram was used to illustrate the fatigue state of the stent while the distribution of the alternating stress points and average stress points are illustrated as in Figure 8. The finite element model simulation of the degradation process was performed on the stent at an equilibrium state after the fatigue analysis, where the changes in the degradation state of the stent and the distribution of the degradation degree were analyzed. Figure 9 illustrates the changes in the degradation degree and the comparison of the degradation degree vs. time at different positions of the stent over 30 days of the degradation process.