For oligonucleotide-directed mutagenesis, equations have been derived to predict how complete a given library is likely to be, and the size of a library required for a given probability of 100% coverage, or other selected threshold. These formulas are widely applied in the design of mutagenesis and screening experiments, but the predictions they generate should be treated cautiously as they are generally based on probabilities of randomly drawing subsets from populations. In practice, every method for generating protein variants has strengths, weaknesses and biases. Thus, ideally parameters describing the performance of mutagenesis methods should be empirically estimated and incorporated into the formulas to avoid underestimation of required screening efforts. The screening is usually the most expensive part of the overall effort, so it is highly desirable to identify the most appropriate screening scale. Thus, the common goal of these procedures is to identify the optimal scale, at which risks of missing positive hits are acceptably low and numbers of clones are not prohibitively high for screening. Frequently, the most important objective is to avoid missing rare “positive hits”. However, in some cases there may be numerous “positive hits”. Thus, retaining variability may be more important and this may have significant implications for mutation generation and screening strategies. b-glucosidase Zm-p60.1 is an enzyme that was originally identified in maize, where it catalyzes the release of active cytokinin from glucoside transport and storage forms, thereby playing a key role in hormonal regulation. Zm-p60.1 is a glucohydrolase of the glycoside hydrolase 1 family. Amino acid residues involved in its substrate interactions and catalysis have been identified using site-directed mutagenesis and X-ray crystallography. A shared feature of GH1 members, and other 8 proteins, 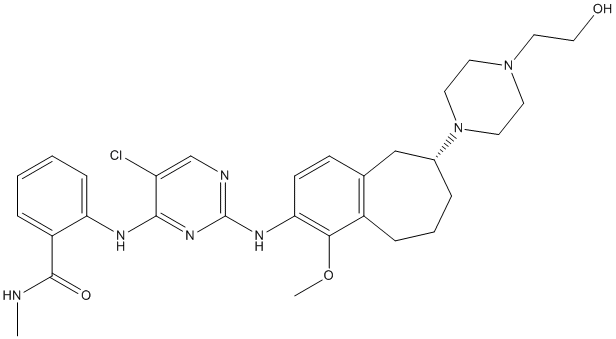 is an active site formed by four variable loops extending from the conserved structure of the protein. A key structural determinant of substrate specificity in Zm-p60.1 �C repeatedly confirmed in structural, docking and kinetic studies �C is the F193�CF200�CW373�CF461 cluster. One of the findings of the cited studies is that W373 plays an important role in stabilizing the aglycone part of the substrate in the entrance of the active site. Residues in Danshensu corresponding positions often play similar roles in other glucosidases, although the amino acid composition is not preserved in all cases. In the presented study we further explored the effects of varying position W373, and unexpectedly found that numerous generated variants had enzymatic activity. This finding led to a detailed consideration of the optimal combination of rational and random strategies for constructing b-glucosidase Zm-p60.1 protein libraries for further structural-functional analyses. Increasing the coverage of variant libraries with a high fraction of positive hits simply by random mutagenesis and/or more extensive screening may be inefficient because the probabilities of detecting variants that have already been detected rather than new ones rapidly increase as libraries Pimozide approach completion. However, knowledge of the theoretical probabilities of finding new variants and the factors influencing the probabilities can greatly facilitate efforts to identify the most appropriate strategy for further screening. Randomized codon-based saturation mutagenesis becomes inefficient for finding.
is an active site formed by four variable loops extending from the conserved structure of the protein. A key structural determinant of substrate specificity in Zm-p60.1 �C repeatedly confirmed in structural, docking and kinetic studies �C is the F193�CF200�CW373�CF461 cluster. One of the findings of the cited studies is that W373 plays an important role in stabilizing the aglycone part of the substrate in the entrance of the active site. Residues in Danshensu corresponding positions often play similar roles in other glucosidases, although the amino acid composition is not preserved in all cases. In the presented study we further explored the effects of varying position W373, and unexpectedly found that numerous generated variants had enzymatic activity. This finding led to a detailed consideration of the optimal combination of rational and random strategies for constructing b-glucosidase Zm-p60.1 protein libraries for further structural-functional analyses. Increasing the coverage of variant libraries with a high fraction of positive hits simply by random mutagenesis and/or more extensive screening may be inefficient because the probabilities of detecting variants that have already been detected rather than new ones rapidly increase as libraries Pimozide approach completion. However, knowledge of the theoretical probabilities of finding new variants and the factors influencing the probabilities can greatly facilitate efforts to identify the most appropriate strategy for further screening. Randomized codon-based saturation mutagenesis becomes inefficient for finding.