This suggests that crisis may promote instability in a restricted region of the genome. Recombination between homologs for sequences that are located proximal to the telomere, and spontaneous translocation formation involving telomere-proximal sequences has been observed in telomerase-deficient yeast cells, we sought to determine the impact of crisis on the rate of spontaneous translocation formation by HR. Spontaneous translocation formation has been shown to occur by HR between dispersed, repetitive genomic sequences in humans, which could be a means of spreading the destabilizing effects of dysfunctional telomeres to regions that do not lie near a telomere. These results demonstrate that spontaneous inter-chromosomal recombination increases during crisis in Atractylenolide-III telomerase deficient cells, but suggests that this increase requires that at least one of the substrates be located proximal to the telomere at the CAN1 locus, where recombinagenic lesions may accumulate during crisis. These results are consistent with the observation of spontaneous translocation formation involving sequences at other telomere proximal loci in telomerase-deficient cells. Effects on heteroallelic recombination have been suggested to be the result of crisis-associated exonucleolytic degradation from uncapped telomeres, which can confer a recombinagenic state on telomere proximal sequences. However, the retention of telomere proximal sequences in the form of a reciprocal translocation chromosome in a significant fraction of the recombinants suggests that crisis-associated translocations need not arise exclusively from exonucleolytic degradation from the telomere. We speculate that DNA replication lesions, such as those thought to stimulate crisis-associated mutagenesis at telomere proximal loci may also contribute to spontaneous translocation formation in senescent cells. Similar events in senescent human cells that have entered crisis could generate translocations by HR between abundant and widely dispersed repetitive sequences, such as Alu elements, which are found near telomeres and can participate in translocation formation by HR. Translocation formation has been shown previously to increase many-fold subsequent to the formation of DSBs adjacent to both recombination substrates, consistent with its occurrence by SSA, a particularly efficient mechanism of HR. In contrast to its effect on spontaneous translocation formation, telomerase deficiency decreased the frequency of DSB-stimulated translocation by as much 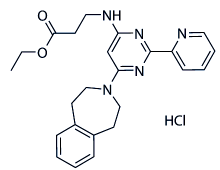 as 250-fold, suggesting that telomerase contributes to DSB repair by HR. Additionally, in telomerase deficient cells the frequency of DSB-stimulated translocation was reduced before, during and after crisis, and regardless of whether either of the recombination substrates was located near a telomere. This suggests that telomerase exerts a genome-wide effect on DSB repair that is independent of its role in forestalling crisis. Analysis of the epistasis interactions between est2D and several HR gene mutations suggested that telomerase plays a role in DSBstimulated translocation formation by SSA that is similar to that of Rad52 but is distinct from it. The observation that telomerase deficiency also reduces translocation formation following the creation of a DSB adjacent to only one recombination Epimedoside-A substrate suggests that telomerase deficiency may have a more general effect on DSB repair by HR. These results are reminiscent of the ionizing radiation sensitivity and DNA repair defects displayed by telomerase deficient mice.
as 250-fold, suggesting that telomerase contributes to DSB repair by HR. Additionally, in telomerase deficient cells the frequency of DSB-stimulated translocation was reduced before, during and after crisis, and regardless of whether either of the recombination substrates was located near a telomere. This suggests that telomerase exerts a genome-wide effect on DSB repair that is independent of its role in forestalling crisis. Analysis of the epistasis interactions between est2D and several HR gene mutations suggested that telomerase plays a role in DSBstimulated translocation formation by SSA that is similar to that of Rad52 but is distinct from it. The observation that telomerase deficiency also reduces translocation formation following the creation of a DSB adjacent to only one recombination Epimedoside-A substrate suggests that telomerase deficiency may have a more general effect on DSB repair by HR. These results are reminiscent of the ionizing radiation sensitivity and DNA repair defects displayed by telomerase deficient mice.