Highly prone to frequent gene rearrangement within a genome and horizontal gene transfer among bacterial and archaeal species. Proteins are often subject to post-translational modification as part of signal transduction pathways to rapidly regulate their activity or abundance in response to changes in the environment. In addition to post-translational modification by small chemical groups such as phosphate, acetyl, or methyl groups, proteins can also be Cinoxacin modified by the Epimedoside-A ubiquitin family of proteins. Ubiquitin is a highly conserved 76-amino acid protein that is covalently attached to substrates either as  single ubiquitin moieties or as ubiquitin chains, with a poly-ubiquitin chain typically targeting its substrate for degradation via the 26S proteasome. Conjugation requires C-terminal processing of the ubiquitin precursor, followed by a cascade of E1, E2, and E3 enzymes that successively activate ubiquitin and direct its attachment to the substrate. Approximately ten other ubiquitin-like proteins, including Small Ubiquitin-like MOdifier, are conjugated to substrates via similar mechanisms, although each pathway has its own dedicated set of enzymes. The functions of SUMO and the other ubiquitin-like proteins are distinct from that of ubiquitin, with frequent cross-pathway regulation or interactions being uncovered. As the final enzyme in the conjugation pathway, the identification of ubiquitin ligase and understanding their substrate specificity, regulation, and functions remains a major goal of ubiquitin research. Three groups of E3s exist, each distinguishable by their sequence motifs and conjugation mechanism. RING and U-box E3s facilitate the transfer of ubiquitin from the E2 to the target by functioning as a scaffold or adaptor that recruits the E2 and substrate together, whereas HECT E3s form a thioester bond with activated ubiquitin, which is subsequently transferred from the HECT active cysteine residue directly onto the target substrate. Most E3s can polymerize chains onto substrates by ligating additional ubiquitin monomers onto any one of seven lysine residues of a previously conjugated ubiquitin. A final class of ubiquitin ligases, E4s, can only ligate ubiquitin onto other ubiquitin residues and therefore requires a pre-conjugated residue to generate a ubiquitin chain on a substrate protein. Ubiquitylation has multiple functions, with its most common role being the targeting of the modified substrate to the proteasome for degradation. For some proteins, however, conjugation to ubiquitin is not sufficient to target them to the proteasome. One of the key intermediaries in the pathway is Cdc48/p97, a highly conserved essential AAA-ATPase that bridges ubiquitylation and proteasomal degradation by using its “segregase” activity to extract a ubiquitylated substrate from membranes or from protein complexes. Cdc48/p97 is required for diverse biological processes including endoplasmic reticulum-associated degradation, processing of the transcription factor Spt23, and the proteolytic ubiquitin fusion degradation and N-end rule pathways. Cdc48 participates in these diverse pathways through its intrinsic ubiquitin-binding activity and by associating with an array of co-factors, including ubiquitin-binding proteins that recruit conjugated proteins to Cdc48, multiple RING finger E3s such as Ufd2, Hrd1, and Doa10, and a deubiquitinating enzyme that regulate chain length. Through this association with a wide variety of co-factors, Cdc48 is involved in a diverse array of cellular processes. The substrate recruitment factors typically bind to the Cdc48 N-terminal domain while the Ufd2 and Ufd3 substrate processing factors bind at the Cdc48 C-terminus.
single ubiquitin moieties or as ubiquitin chains, with a poly-ubiquitin chain typically targeting its substrate for degradation via the 26S proteasome. Conjugation requires C-terminal processing of the ubiquitin precursor, followed by a cascade of E1, E2, and E3 enzymes that successively activate ubiquitin and direct its attachment to the substrate. Approximately ten other ubiquitin-like proteins, including Small Ubiquitin-like MOdifier, are conjugated to substrates via similar mechanisms, although each pathway has its own dedicated set of enzymes. The functions of SUMO and the other ubiquitin-like proteins are distinct from that of ubiquitin, with frequent cross-pathway regulation or interactions being uncovered. As the final enzyme in the conjugation pathway, the identification of ubiquitin ligase and understanding their substrate specificity, regulation, and functions remains a major goal of ubiquitin research. Three groups of E3s exist, each distinguishable by their sequence motifs and conjugation mechanism. RING and U-box E3s facilitate the transfer of ubiquitin from the E2 to the target by functioning as a scaffold or adaptor that recruits the E2 and substrate together, whereas HECT E3s form a thioester bond with activated ubiquitin, which is subsequently transferred from the HECT active cysteine residue directly onto the target substrate. Most E3s can polymerize chains onto substrates by ligating additional ubiquitin monomers onto any one of seven lysine residues of a previously conjugated ubiquitin. A final class of ubiquitin ligases, E4s, can only ligate ubiquitin onto other ubiquitin residues and therefore requires a pre-conjugated residue to generate a ubiquitin chain on a substrate protein. Ubiquitylation has multiple functions, with its most common role being the targeting of the modified substrate to the proteasome for degradation. For some proteins, however, conjugation to ubiquitin is not sufficient to target them to the proteasome. One of the key intermediaries in the pathway is Cdc48/p97, a highly conserved essential AAA-ATPase that bridges ubiquitylation and proteasomal degradation by using its “segregase” activity to extract a ubiquitylated substrate from membranes or from protein complexes. Cdc48/p97 is required for diverse biological processes including endoplasmic reticulum-associated degradation, processing of the transcription factor Spt23, and the proteolytic ubiquitin fusion degradation and N-end rule pathways. Cdc48 participates in these diverse pathways through its intrinsic ubiquitin-binding activity and by associating with an array of co-factors, including ubiquitin-binding proteins that recruit conjugated proteins to Cdc48, multiple RING finger E3s such as Ufd2, Hrd1, and Doa10, and a deubiquitinating enzyme that regulate chain length. Through this association with a wide variety of co-factors, Cdc48 is involved in a diverse array of cellular processes. The substrate recruitment factors typically bind to the Cdc48 N-terminal domain while the Ufd2 and Ufd3 substrate processing factors bind at the Cdc48 C-terminus.
Author: screening library
Direct evidence about the ability of T cell-activated macrophages in specific recognition and rejection
These activated macrophages Butenafine hydrochloride produce excessive amounts of NO which has cytotoxic and cytostatic effects and causes direct tissue damage. Several studies provided indirect and direct evidence for the involvement of NO in graft rejection. A significant increase in the iNOS expression and NO production has been reported in acute graft rejection. Vos et al. study also revealed that administration of iNOS blockers after transplantation improves renal allograft function and reduces graft injury. In this study we demonstrated that peritoneal macrophages, treated with the IDO-expressing fibroblast conditioned medium, shows Ergosterol significantly lower levels of iNOS expression in response to LPS+IFN-c stimulation. We also showed that tryptophan deficiency rather than kynurenine enrichment inhibits iNOS expression and NO production by stimulated Raw264.7 cells. The specific inhibitory effect of tryptophan deficiency on iNOS expression and NO production by activated macrophages was further confirmed by the restored macrophage proinflammatory activity in response to tryptophan supplementation to the Trp-D medium. In 1997 Sekkai et al. demonstrated the inhibitory effect of 3-hydroxyanthranilic acid, a kynurenine pathway metabolite, on iNOS expression and enzymatic activity via inhibition of NF-kB activation. They also showed that other tryptophan metabolites including kynurenine have no significant inhibitory effect on NF-kB activation which support our data related to the noninhibitory effect of kynurenine on iNOS expression and NO production. This study provides evidence for inhibitory effects of tryptophan deprivation on macrophages proliferation, survival and proinflammatory activity. In order to evaluate the immunoprotective effects of local IDO expression, xenografts, rat islets imbedded in an IDO expressing fibroblast populated matrix, were transplanted in B6 mice. Using IDO expressing fibroblast we were able to demonstrate that local IDO expression inhibits the infiltration of CD3 + T cells into the xenogeneic islet 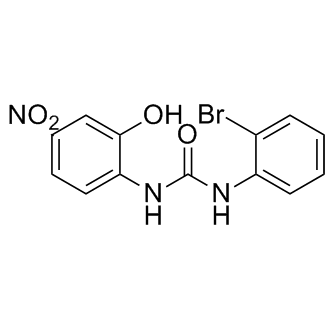 graft. This is in agreement with other studies using stable IDO expressing cell line or IDO expressing bystander fibroblasts. Additionally as it is shown in this study, local IDO expression by bystander fibroblasts significantly inhibits the infiltration of macrophages into the islet xenografts and impairs their proinflammatory responses. Keeping in mind that kidney capsule is a confined micro-environment, the high levels of IDO expression by fibroblast-populated islet composite leads to a remarkable reduction in the tryptophan concentration and kynurenines accumulation in vivo at the graft site. While not negatively affecting the islet function, this low-tryptophan microenvironment protect islet xenografts by inhibiting the infiltration, proliferation and inflammatory activity of immune cells. In conclusion this study is the first to demonstrate modulation of the innate immune response by IDO as a result of creating a tryptophan deficient environment. Specifically we have shown that IDO immunomodulatory effects might be mediated via inhibition of proliferation, induction of apoptosis or impairment of proinflammatory responses in macrophages. Our In vivo study demonstrated that macrophage and T cell infiltration into a xenogenic islet graft is significantly reduced in IDO expressing grafts and iNOS expression is inhibited in these infiltrating macrophages. Data generated from this study along with those previously reported by our group support the feasibility and applicability of using IDO expression as a local immunosuppressive strategy for protecting both allo- and xeno islet grafts from immune rejection.
graft. This is in agreement with other studies using stable IDO expressing cell line or IDO expressing bystander fibroblasts. Additionally as it is shown in this study, local IDO expression by bystander fibroblasts significantly inhibits the infiltration of macrophages into the islet xenografts and impairs their proinflammatory responses. Keeping in mind that kidney capsule is a confined micro-environment, the high levels of IDO expression by fibroblast-populated islet composite leads to a remarkable reduction in the tryptophan concentration and kynurenines accumulation in vivo at the graft site. While not negatively affecting the islet function, this low-tryptophan microenvironment protect islet xenografts by inhibiting the infiltration, proliferation and inflammatory activity of immune cells. In conclusion this study is the first to demonstrate modulation of the innate immune response by IDO as a result of creating a tryptophan deficient environment. Specifically we have shown that IDO immunomodulatory effects might be mediated via inhibition of proliferation, induction of apoptosis or impairment of proinflammatory responses in macrophages. Our In vivo study demonstrated that macrophage and T cell infiltration into a xenogenic islet graft is significantly reduced in IDO expressing grafts and iNOS expression is inhibited in these infiltrating macrophages. Data generated from this study along with those previously reported by our group support the feasibility and applicability of using IDO expression as a local immunosuppressive strategy for protecting both allo- and xeno islet grafts from immune rejection.
High protein solubility and stability of camelid VHH domains are likely to help their effective translocation
In previous works, we have Epimedoside-A studied the secretion mechanism of ATs and reported their capacity to translocate a model sdAb to the surface of E. coli fused to the ��-domain of ATs such as the IgA protease from Neisseria gonorrhoeae and EhaA from enterohemorrhagic E. coli O157:H7. A model sdAb fused to these ��-domains was correctly folded in the periplasm with the canonical disulfide bond of Ig domains formed by the action of DsbA. In a different study, we demonstrated that the Intimins from EHEC and enteropathogenic E. coli could be expressed in E. coli K-12 and displayed their native passenger domains on the cell surface with a disulfide bond formed by DsbA. However, neither the display of heterologous Ig domains fused to Intimin, nor the utility of the ��-domains of ATs and Intimin for display of sdAb libraries and de novo selection of sdAbs against an antigen of interest was investigated. In this work, we have demonstrated the capacity of the ��domains of EhaA and Intimin for the display of sdAb libraries on the surface of E. coli cells and for the selection of novel sdAbs against the extracellular domain of the translocated intimin receptor from EHEC. E. coli display libraries were screened by magnetic cell sorting and clones that specifically bound TirMEHEC were isolated, characterized by flow cytometry, as well as by enzyme linked immunosorbent assay and surface plasmon resonance with the purified sdAb. In addition, E. coli display was used to provide an estimation of the affinity of the selected sdAb by flow cytometry analysis under equilibrium conditions before protein purification. In this work, we have demonstrated that the ��-domains of EhaA and Intimin from EHEC O157:H7 are effective platforms for the display sdAb libraries on the surface of E. coli K-12 cells, and allow the selection of high affinity sdAbs from immune libraries using biotinylated antigen and MACS. Despite their opposite topologies, both systems express stable fusion proteins with the native ��-barrel correctly folded in the OM and display a functional sdAb with antigen-binding capacity on the surface of E. coli cells. Most sdAbs in the immune library were displayed on E. coli at good expression levels with both CEhaA and Neae, with an average between 6000-8000 molecules/bacterium. From the immune libraries constructed in our study, we obtained a total of five independent camelid VHH sequences binding the antigen used in the immunization, being the sdAb of higher affinity and specificity the more frequent clone found in the selections with both ��-domains. These results demonstrate that both E. coli display systems can be used to retrieve high-affinity Ginsenoside-F4 binders from immune libraries of camelid VHH sequences. Fewer different binders were found compared to other reported studies using an immune library of camelid VHH displayed on the surface of phages and Staphyloccous carnosus cells. However, we think that the relatively small number of binders obtained in our study reflects a limitation of the anti-TirMEHEC library employed, and not of the E. 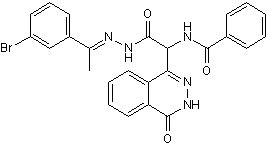 coli-display systems described. Firstly, we have performed selections of the antiTirMEHEC VHH library on phage that failed to retrieve other specific binders than those selected by E. coli-display. Secondly, ongoing work in our laboratory with other immune VHH libraries also seem to indicate that essentially the same pool of binders can be isolated from phage and E. colidisplay systems. Thus, it appears that rather than the display system used, the actual diversity of binders retrieved from an immune VHH library is more dependent on factors such as immunogenicity of the antigen, number of animals used in the immunization, and library size.
coli-display systems described. Firstly, we have performed selections of the antiTirMEHEC VHH library on phage that failed to retrieve other specific binders than those selected by E. coli-display. Secondly, ongoing work in our laboratory with other immune VHH libraries also seem to indicate that essentially the same pool of binders can be isolated from phage and E. colidisplay systems. Thus, it appears that rather than the display system used, the actual diversity of binders retrieved from an immune VHH library is more dependent on factors such as immunogenicity of the antigen, number of animals used in the immunization, and library size.
Facilitated by the addition of specific ligands or inhibitors which bind to the protein and lower its intrinsic heterogeneity
Finally, the addition of trace amounts of protease to the crystallization trials �C in situ proteolysis �C rescued 10 out of 69 different proteins that had previously failed in crystallization and structure determination. In situ proteolysis appears to be the most efficacious crystallization rescue strategy. However, while the study of Dong et al. was systematic and rigorous, all but one of the successful cases derived from a single experimenter, and the proteins were predominantly of bacterial origin. This prompted further inquiry into whether the Mechlorethamine hydrochloride method would be applicable to human proteins, would be as successful in other hands, and would be useful for those proteins for which dozens of variants had already been tested. This paper describes an expanded study: applying in situ proteolysis to 270 new proteins since the last paper, from prokaryotes and eukaryotes, and by dozens of scientists. The intent of Dong et al. was to carry out a systematic, statistically significant, and well-controlled test of the efficacy of in situ proteolysis. Each new crystal was analyzed using mass spectrometry to ensure that the success of the method could be attributed directly to the use of proteases. Their paper strongly suggested that the method should be adopted as a primary crystallization strategy due to its high success rate, but was qualified by the fact that the method was being employed in a very controlled setting. The intent of this study was to examine the efficacy of the method in practice, as carried out on a larger number of proteins by a larger number of experimenters. This strategy had the advantage of exploring the use of the method under less controlled conditions, in which individual investigators adopted slightly different methodologies and strategies. The disadvantage of this strategy is that any conclusions drawn have more  caveats, due to the inability to control all aspects of the Acetylcorynoline experiments. We describe here several variations to the original method that have proven successful. Our default strategy for human proteins is to design 10�C15 different constructs for each protein and to attempt to crystallize each one that can be purified, in both the presence and absence of the histidine tag. If this first round fails, more constructs with slight variations at the N- and C-terminal positions are often created, though this strategy is met with significantly diminishing returns. In situ proteolysis is now being used to resuscitate some of the failed projects. In one case, 36 constructs were purified for the histone methyltransferase SETDB1 protein, and none crystallized, either with or without the histidine tag. In comparison with protein antibodies, nucleic acid aptamers are more easily obtained, modified and manipulated. The specific targets recognized by aptamers cover metal ions, small molecules, proteins, and even whole cells or viruses. These unique properties endow aptamers with great application potential in clinical therapeutics, biological researches, molecular recognition, bioanalysis and sensing. This is attracting more and more efforts directed to the development of new functional aptamers, such as catalytic aptazymes consisting of an aptamer domain and a ribozyme module. Because of their enzyme functions, aptazymes are widely applied to the detection of various targets that specifically bind to the aptamer domain and, in general, cause a conformational change. Interestingly, a few specific DNAs are able to serve as both aptamers and ribozymes.
caveats, due to the inability to control all aspects of the Acetylcorynoline experiments. We describe here several variations to the original method that have proven successful. Our default strategy for human proteins is to design 10�C15 different constructs for each protein and to attempt to crystallize each one that can be purified, in both the presence and absence of the histidine tag. If this first round fails, more constructs with slight variations at the N- and C-terminal positions are often created, though this strategy is met with significantly diminishing returns. In situ proteolysis is now being used to resuscitate some of the failed projects. In one case, 36 constructs were purified for the histone methyltransferase SETDB1 protein, and none crystallized, either with or without the histidine tag. In comparison with protein antibodies, nucleic acid aptamers are more easily obtained, modified and manipulated. The specific targets recognized by aptamers cover metal ions, small molecules, proteins, and even whole cells or viruses. These unique properties endow aptamers with great application potential in clinical therapeutics, biological researches, molecular recognition, bioanalysis and sensing. This is attracting more and more efforts directed to the development of new functional aptamers, such as catalytic aptazymes consisting of an aptamer domain and a ribozyme module. Because of their enzyme functions, aptazymes are widely applied to the detection of various targets that specifically bind to the aptamer domain and, in general, cause a conformational change. Interestingly, a few specific DNAs are able to serve as both aptamers and ribozymes.
Raised about the relevance of psychopharmacological prescriptions in primary health care practice
These drugs can have serious side effects and some of them, especially the benzodiazepines have an addictive potential. Therefore, their prescription and use should be monitored by a healthcare professional. Also, the treatment of mental disorders entails more than just prescription of medicines; it also requires the doctor to develop a strong bond with the patient, and to counsel the patients regarding alternative treatment options. In this context, several of our findings were distressing. Almost 51% of the patients with psychopharmacological medications did not know their diagnosis. Several factors could be responsible for this. Most of the psychopharmacological medicines in our study were antidepressants and it is possible that doctors were hesitant to tell patients that they were suffering from depression. Further, patients may have refused the diagnosis due to the social stigma associated with psychiatric diseases, or they may have been unable to understand the diagnosis due to Pancuronium dibromide cognitive disability associated with psychiatric illnesses. At the same time it can also be argued that the doctors were unable to effectively explain the diagnosis to their patients because they themselves were unclear about it. With almost 71% of the patients no alternative treatment options were discussed. This issue must be addressed because addition of non-pharmacological treatment options can be helpful in augmenting the treatment of common mental disorders like anxiety and Butenafine hydrochloride depression, and in milder cases these may be the only forms of treatment required. This finding can also be interpreted as a sign of over dependence on medications on the part of family physicians when it comes to managing such disorders, given that often there are several pharmacological and non-pharmacological treatment options. Adverse effects of the drugs had not been explained to almost 86% of the patients being prescribed psychopharmacological medications. SSRIs have fewer side effects, but TCAs and benzodiazepines have significant ill-effects which should be discussed with the patients before initiating therapy. In general, most patients attending primary care services want to know the adverse effects of the treatment being prescribed to them. Although, most patients knew about the duration of treatment, a third did not. This can prove hazardous as prolonged treatment with benzodiazepines can lead to dependence, while stopping antidepressants before time is likely to lead to relapse. With almost 82% of the patients no referral plan was discussed. This complements the findings of a previous study which showed that only 3% of the patients in a psychiatry clinic had been referred by primary care physicians. The low rate of referral makes it all the more important for the physicians at 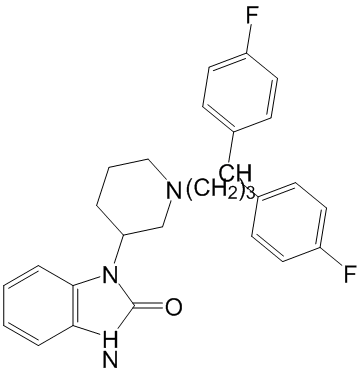 first level of care to be well versed in the management of mental illness. In a developing country like Pakistan mental illnesses make up a significant proportion of the total disease burden and trained psychiatrists are few. Consequently, correct identification and effective management of common psychiatric disorders by family physicians becomes essential. The most common psychopharmacological medicines prescribed were antidepressants with relatively fewer prescriptions of benzodiazepines. This pattern differs from other studies which show benzodiazepines rather than antidepressants as being the most commonly prescribed psychopharmacological medicine. This is an encouraging finding as benzodiazepines have a high potential for addiction and should be used.
first level of care to be well versed in the management of mental illness. In a developing country like Pakistan mental illnesses make up a significant proportion of the total disease burden and trained psychiatrists are few. Consequently, correct identification and effective management of common psychiatric disorders by family physicians becomes essential. The most common psychopharmacological medicines prescribed were antidepressants with relatively fewer prescriptions of benzodiazepines. This pattern differs from other studies which show benzodiazepines rather than antidepressants as being the most commonly prescribed psychopharmacological medicine. This is an encouraging finding as benzodiazepines have a high potential for addiction and should be used.