Since proteasome inhibitors down-regulate FoxM1 and FoxM1 overexpression protects against cell death mediated by proteasome inhibitors, suppression of FoxM1 may be required for the anticancer activity of these drugs. Further experiments are needed to clarify the mechanisms of FoxM1 down-regulation by proteasome inhibitors and the importance of FoxM1 suppression in the activity of proteasome inhibitors as anticancer drugs. Angiogenesis, the sprouting of new vessels from the existing vasculature, mainly occurs during embryonic development and growth. In the adult it is restricted to distinct physiological processes, e.g. wound healing, by a balance of pro- and antiangiogenic factors. Unregulated angiogenesis is one of the hallmarks of 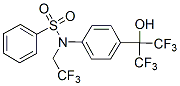 cancer. Tumor growth is highly dependent on proper supply with oxygen and nutrients and removal of metabolic waste. Therefore, angiogenesis is crucial for tumor survival and proliferation, and tumor size remains limited unless the tumor switches to an angiogenic phenotype. The intent to stop tumor growth and finally starve the tumor by disrupting angiogenic signaling has led to the development of anti-angiogenic drugs for anticancer therapy. Agents addressing vascular endothelial growth factorinduced angiogenesis have already been successfully introduced into tumor therapy. However, in clinical use it has become apparent that antiangiogenic tumor therapy is more challenging than expected: Many tumors are refractory to VEGF-blockade or become resistant during treatment. This evasive resistancecan be caused by a shift to alternative angiogenic signaling pathways due to a pre-existing multiplicity of redundant pro-angiogenic signals. Therefore novel targets in angiogenesis need to be identified and characterized as a basis for future INCB28060 therapeutic concepts. Cdk5 has been discovered as a neuronal cdc2-like serine/ threonine kinasein 1992. Despite its high sequence homology with the mitotic Cdk1, Cdk5 is not involved in cell cycle control and unique among the Cdks in its regulation and function. On the cellular level, Cdk5 is well-described in neurons as the key hub in the dynamic network of trafficking and transport, integrating signals in cytoskeletal dynamics during neuronal migration, in synaptic plasticity and synaptic vesicle endo- and exocytosis, cell adhesion and axon guidance, neuromuscular development and pain signaling. Although Cdk5 expression and activity is highest in the central nervous system, Cdk5 is also expressed in various tissues, and an increasing body of research uncovers extraneuronal functions of Cdk5, where it is involved in the regulation of migration, cell death and survival, glucose metabolism and inflammation. -roscovitine or CYC-202/seliciclib �C in the following referred to as roscovitine �C belongs to the class of 2,6,9-trisubstituted purines. It is one of the best-known Cdk inhibitors, and is currently tested in several phase I and phase II clinical trials for tumor treatment. Roscovitine inhibits mainly Cdk1, Cdk2, Cdk5, Cdk7 and Cdk9 and exerts anti-mitotic and Foretinib pro-apoptotic effects in a wide range of tumor cells. Cell-cycle independent actions of roscovitine mainly derive from Cdk5 inhibition and include anti-angiogenicand anti-inflammatoryeffects, inhibition of cell migration and motilityand modulation of glucose metabolism. Anti-angiogenic actions of Cdk inhibitors have been observed in vitro and in vivo. Recently, we have demonstrated that the anti-angiogenic effect of roscovitine most likely results from impaired endothelial cell migration. The effect on migration was traced down to Cdk5 inhibition which led to Rac1 inactivation and lamellipodia disruption.
cancer. Tumor growth is highly dependent on proper supply with oxygen and nutrients and removal of metabolic waste. Therefore, angiogenesis is crucial for tumor survival and proliferation, and tumor size remains limited unless the tumor switches to an angiogenic phenotype. The intent to stop tumor growth and finally starve the tumor by disrupting angiogenic signaling has led to the development of anti-angiogenic drugs for anticancer therapy. Agents addressing vascular endothelial growth factorinduced angiogenesis have already been successfully introduced into tumor therapy. However, in clinical use it has become apparent that antiangiogenic tumor therapy is more challenging than expected: Many tumors are refractory to VEGF-blockade or become resistant during treatment. This evasive resistancecan be caused by a shift to alternative angiogenic signaling pathways due to a pre-existing multiplicity of redundant pro-angiogenic signals. Therefore novel targets in angiogenesis need to be identified and characterized as a basis for future INCB28060 therapeutic concepts. Cdk5 has been discovered as a neuronal cdc2-like serine/ threonine kinasein 1992. Despite its high sequence homology with the mitotic Cdk1, Cdk5 is not involved in cell cycle control and unique among the Cdks in its regulation and function. On the cellular level, Cdk5 is well-described in neurons as the key hub in the dynamic network of trafficking and transport, integrating signals in cytoskeletal dynamics during neuronal migration, in synaptic plasticity and synaptic vesicle endo- and exocytosis, cell adhesion and axon guidance, neuromuscular development and pain signaling. Although Cdk5 expression and activity is highest in the central nervous system, Cdk5 is also expressed in various tissues, and an increasing body of research uncovers extraneuronal functions of Cdk5, where it is involved in the regulation of migration, cell death and survival, glucose metabolism and inflammation. -roscovitine or CYC-202/seliciclib �C in the following referred to as roscovitine �C belongs to the class of 2,6,9-trisubstituted purines. It is one of the best-known Cdk inhibitors, and is currently tested in several phase I and phase II clinical trials for tumor treatment. Roscovitine inhibits mainly Cdk1, Cdk2, Cdk5, Cdk7 and Cdk9 and exerts anti-mitotic and Foretinib pro-apoptotic effects in a wide range of tumor cells. Cell-cycle independent actions of roscovitine mainly derive from Cdk5 inhibition and include anti-angiogenicand anti-inflammatoryeffects, inhibition of cell migration and motilityand modulation of glucose metabolism. Anti-angiogenic actions of Cdk inhibitors have been observed in vitro and in vivo. Recently, we have demonstrated that the anti-angiogenic effect of roscovitine most likely results from impaired endothelial cell migration. The effect on migration was traced down to Cdk5 inhibition which led to Rac1 inactivation and lamellipodia disruption.